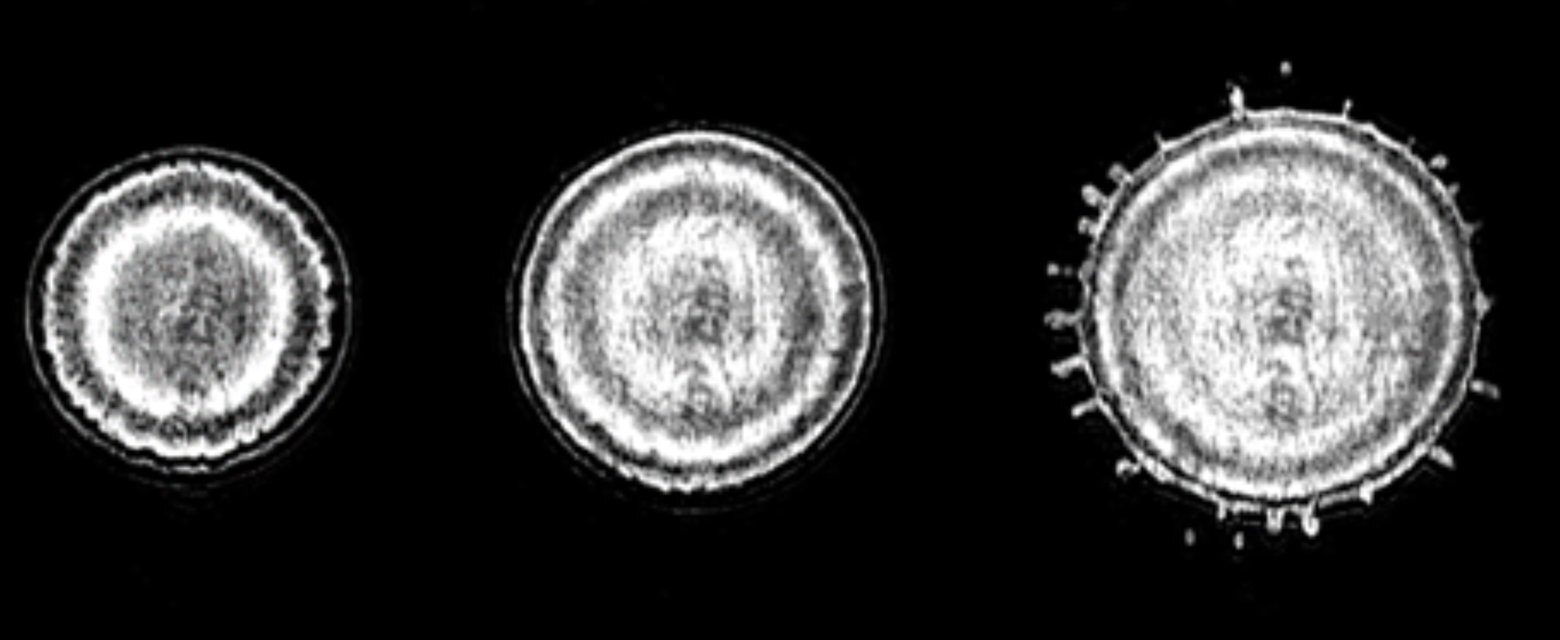
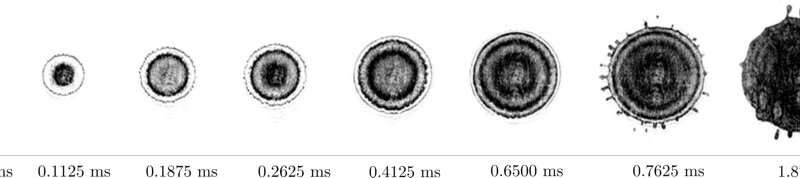
When rain falls on a surface that is still frozen, it makes the road very slippery in a short time. This is an example of liquid droplets falling on a surface that has a temperature below the melting point — it is "supercooled." The freezing of the droplet and crystallization evoke the star-shaped dendritic structures often observed in snowflakes. If the surface is colder, however, the droplet not only freezes faster, but the mechanism changes, as well. At a surface that is cold enough, a remarkable phenomenon occurs: From the center of the droplet, ice fronts move toward the edge while the droplet is still spreading. This happens repeatedly, until the droplet is fully frozen.
Filming from below
The UT researchers observed this by filming the freezing of the droplet from below, exactly at the surface. Laser light is reflected at the interface and filmed using a high-speed camera. This is also called total internal reflection (TIR), and is based on the same method that is used for taking fingerprints. In the experiments, the falling droplet is of hexadecane, which has a melting point of 18 degrees Celsius. The waves were observed when the surface temperature was lowered to 11 degrees below this point.
Internal flow
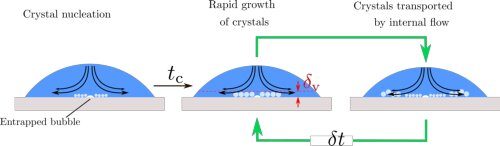
In their theoretical explanation in PNAS, the UT scientists show that the droplet is coldest at the point of impact, that is, in the middle. Crystals form around this, but at the same time, the internal fluid flow pushes them to the boundaries. This process keeps repeating itself until the whole droplet freezes. The study also reveals that the temperature of the surface changes the way the solidified droplet attaches to the surface, thus changing the ease at which it can be "peeled off."
The research not only gives fundamental insight into the process of freezing, it could help researchers developing anti-icing surfaces like those for airplanes. It can improve 3-D printing techniques that make use of solidification of molten wax. And it can help advance extreme ultraviolet lithography (EUV) for chip manufacturing. There, molten metallic droplets that solidify on mirrors could obstruct the whole process.
The paper, "Fast-freezing kinetics inside a droplet impacting on a cold surface," is published in the Proceedings of the National Academy of Sciences (PNAS).
Physics of Fluids - droplet on cold surface 1 from University of Twente on Vimeo.
More information: Pallav Kant et al. Fast-freezing kinetics inside a droplet impacting on a cold surface, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.1912406117
Journal information: Proceedings of the National Academy of Sciences



Comment: See also: