
© Fratic00/shutterstockAt the scale of the very small things are both particles and waves, and the wave nature can cause quantum interference, something that has been found even in very simple chemical reactions, such as between a hydrogen atom and a hydrogen-deuterium molecule.
Scientists thought that even with all the complexity and strangeness quantum mechanics involves, they at least had a good grasp of the simplest chemical reactions, those involving very few electrons and protons. Further investigation showed that's not really the case, confirming physicists' rueful observation that no matter how weird you think quantum behavior might be, the reality will always out-weird you.
Hydrogen has the simplest atom there is, just one proton and electron, followed by its isotope deuterium, which has the same pair and a neutron as well. So when a hydrogen atom meets a molecule made of up hydrogen and deuterium, and replaces the deuterium (H + HD → H
2 + D in chemistry notation), there are not a lot of moving parts - three protons, three electrons, and a neutron. Compared to most chemical reactions, this is about as simple as it gets. As a result, the reaction has been studied intensely as a gateway to greater complexity.
However, a team from the Dalian Institute of Chemical Physics have reported in
Science that
a form of quantum interference occurs during the course of this reaction. Not only has this been missed in previous studies of hydrogen/deuterium displacement, it's never been seen before at all.
The authors report that
when they crossed a beam of hydrogen atoms with another of hydrogen/deuterium molecules, the hydrogen could displace deuterium in two different ways. The first method involves the incoming hydrogen atom colliding with the hydrogen part of the molecule, knocking it out and the two going off together. In the second case, the incoming atom causes the bond between the existing hydrogen and deuterium atoms to stretch, at which point it inserts itself inside.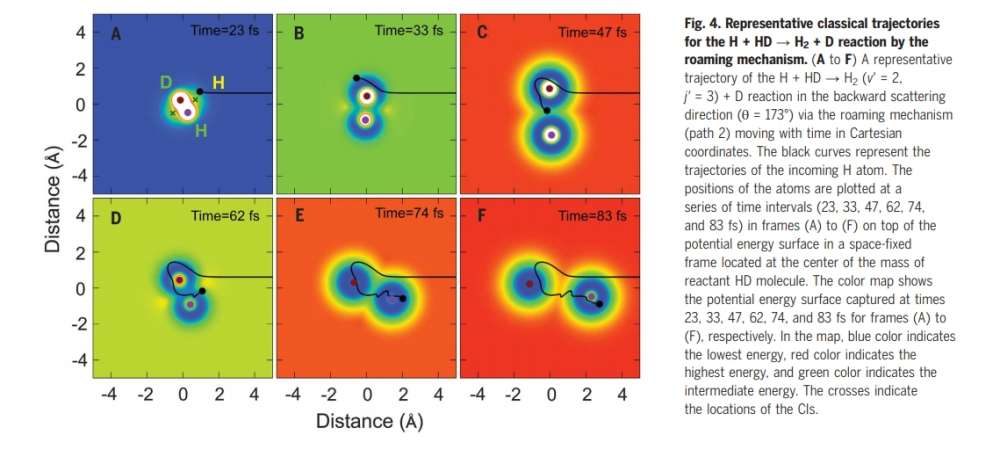
© Xie et al/ScienceThe second, rarer approach, by which a hydrogen atom will displace deuterium from a molecule where it was bonded with another hydrogen atom.
One might think of this like a single person seeking to break up a couple and marry one of the partners. They might sweep one partner off their feet, carrying them away before the jilted lover knows what is happening. Alternatively, the interloper may hang around creating stress within the relationship until the previous closeness loosens and they can form a new bond instead.
In a classical (that is non-quantum) situation, one or another of these would occur. However, at the heart of quantum mechanics is that sometimes particles can do two apparently incompatible things at once, most famously a photon going through two slits simultaneously. We know this happens when the two states created interfere with each other.
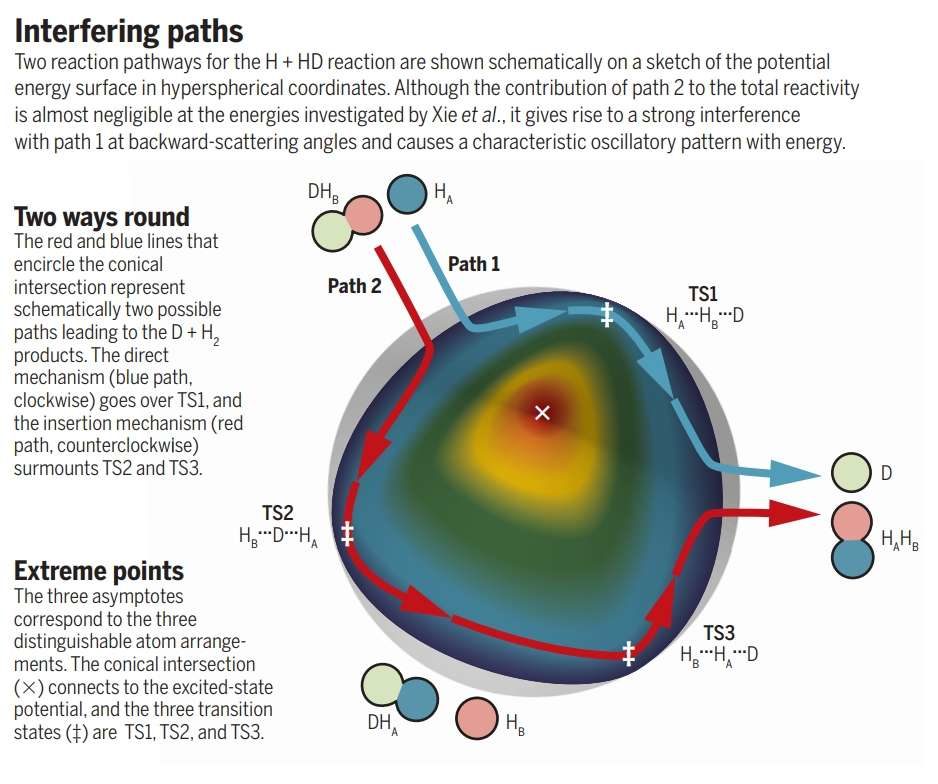
© Aoiz/ScienceIrrespective of the path by which hydrogen displaces deuterium, the molecule and displaced atom go off in the same direction, but with oscillations that prove quantum interference is occurring.
and colleagues observed something very similar.
The incoming hydrogen atoms displaced the deuterium in both ways at once (although the first method was dominant). The waveform created by the different methods interfered with itself, creating distinctive oscillations in the newly formed molecules that allowed the researchers to know both events had happened.
And you thought your love life was complicated.






There was a recent article in Sott about the intelligence in cells and it is because of this requirement of the laws. Intelligence is inherent in the design. These same rules are what prevent a commingling of humans and ants for example, or humans and dogs. Horses can breed with donkeys, producing mules ( one of few commingles allowed) but the mules can not then pro-create. Every species has its own rules about how it procreates and rules preventing it from procreating with other species.
As humans we tend to want to develop along our own path and then impose our creation on the natural order without first understanding the natural order. GMO is an example and glyphosates - they destroy the soil which was designed to produce our food. The creation of plastics another; now they pollute all the waterways of the planet affecting all marine species. Also pharmaceuticals and vaccines which diminish the immune system we were provided. I could go on.
We need to discover a lot more about the natural systems created on earth to support our life, before we continue on this path of destruction. It is our arrogant belief that we know best.