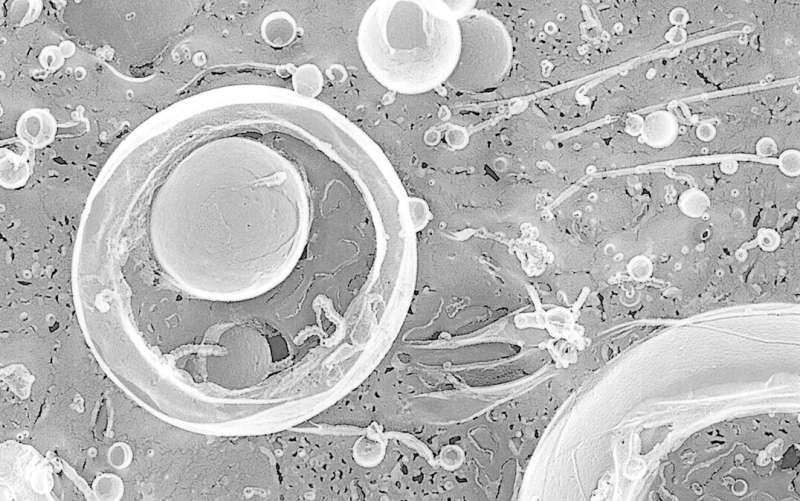
© University of TsukubaCorynebacterium glutamicum.
Researchers from the University of Tsukuba identified a novel mechanism by which bacteria form membrane vesicles, which bacteria employ to communicate with each other or to defend themselves against antibiotics. By studying mycolic acid-containing bacteria (MCB), which also includes tuberculosis-causing bacteria, the researchers demonstrated that environmental stimuli dictate the route by which the MCB form membrane vesicles. Further, their observations were consistent among various MCB. This study has implications for vaccine development as well as novel therapies.
Bacteria have the ability to form membrane vesicles to communicate with each other, but also to defend themselves against antibiotics. In a new study, researchers from the University of Tsukuba discovered a novel mechanism by which mycolic acid-containing bacteria, a specific group of bacteria with a special type of cell membrane, form membrane vesicles.
Bacteria have traditionally been classified on the basis of the composition of their cell envelopes. For example, microbiologists employ Gram staining to differentiate between bacteria that have a thick (Gram-positive) or thin (Gram-negative) cell wall. While bacterial membranes mostly act as protective barriers, they can also form protrusions to make membrane vesicles with diverse biological functions. For example, membrane vesicles may contain various biomolecules, such as DNA, which can be sent between bacteria and confer new abilities to the cells. Membrane vesicles have also been shown to be an important tool for bacteria to defend themselves against antibiotics and phages (viruses that infect bacteria). Recent studies have shown that bacteria form membrane vesicles in various ways, which in turn produces different types of membrane vesicles. While these studies mostly explored the biogenesis of membrane vesicles in Gram-negative and Gram-positive bacteria, the mechanism of membrane vesicle formation in mycolic acid-containing bacteria (MCB), such as Mycobacteria tuberculosis that are responsible for tuberculosis, has remained unknown.
"Mycolic acid-containing bacteria are a very interesting group of bacteria because of their complex cell structure," says author of the study Dr. Toshiki Nagakubo. "The goal of our study was to understand how these cells form membrane vesicles."
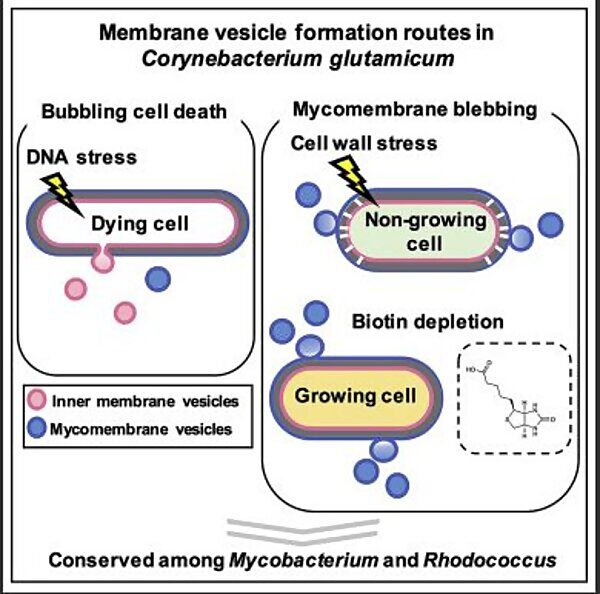
Graphical abstract of vesicle formation pathways
To achieve their goal, the researchers asked how environmental conditions influence the formation of membrane vesicles. They exposed the MCB Corynebacterium glutamicum to two different types of stress: DNA damage and envelope stress, that is an interference with cell wall or cell membrane synthesis. By employing electron microscopy, super resolution live-cell imaging and various biochemical analytical tools, the researchers found that under DNA-damaging conditions, MCB formed membrane vesicles with more diverse morphologies than under normal conditions, demonstrating how bacteria adapt and respond to their environment. They further showed that DNA damage induced membrane vesicle formation remarkably through cell death. On the contrary, exposing the bacteria to envelope stress via penicillin or biotin deficiency resulted in membrane vesicle formation through membrane blebbing. Interestingly, these various routes of membrane vesicle formation were similar in other MCB, demonstrating how the complex cell structure of MCB dictates the types of membrane vesicles this group of bacteria can form.
"These are striking results that provide insight into the mechanisms by which unicellular organisms, namely bacteria, form various types of membrane vesicles. These findings could be helpful for the development of novel therapeutics or vaccines," says corresponding author of the study Associate Professor Masanori Toyofuku.
More information: Toshiki Nagakubo, Yuhei O. Tahara, et al., "Mycolic acid-containing bacteria trigger distinct types of membrane vesicles through different routes",
Cell Volume 24, ISSUE 1,
https://doi.org/10.1016/j.isci.2020.102015
Reader Comments