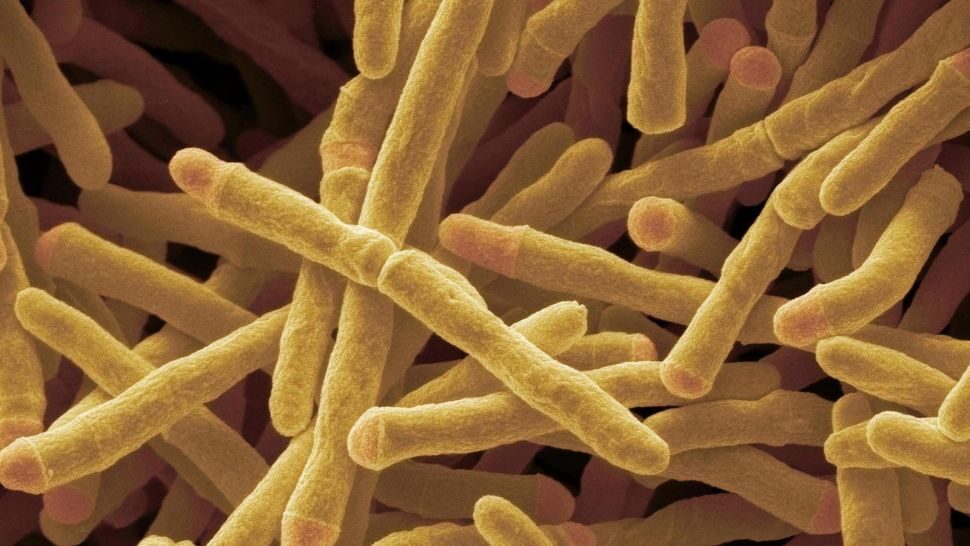
The enzyme, which has been named Huc, is used by the bacterium Mycobacterium smegmatis to draw energy from atmospheric hydrogen, enabling it to survive in extreme, nutrient-poor environments.
Now, by extracting and studying the enzyme, the researchers say they have found a new energy source that could be used to power a range of small portable electrical devices. They published their findings March 8 in the journal Nature.
"We imagine that a Huc-containing power source could power a range of small portable devices using air, including biometric sensors, environmental monitors, digital clocks, and calculators or simple computers," lead author Rhys Grinter, a microbiologist at Monash University in Australia, told Live Science via email.
"When you provide Huc with more concentrated hydrogen, it produces more electrical current," he said. "Which means you could use it in fuel cells to power more complex devices, like smart watches, or smartphones, more portable complex computers, and possibly even a car."
M. smegmatis is a nonpathogenic, fast-growing bacterium often used in the lab to study the cell wall structure of its close, disease-causing relative, Mycobacterium tuberculosis. Commonly found in soil all over the world, M. smegmatis has long been known to convert trace hydrogen in the air into energy; in this way, the microbe can survive in the toughest environments, including Antarctic soils, volcanic craters and the deep ocean, where little other fuel can be found, the researchers said.
But until now, how M. smegmatis did this was a pervading mystery.
To investigate the chemistry behind M. smegmatis' shocking ability, the scientists first isolated the Huc enzyme responsible for the process using chromatography — a lab technique which enables scientists to separate the components of a mixture. Then, they investigated the enzyme's atomic structure with cryo-electron microscopy, a technique that won its creators the 2017 Nobel Prize in chemistry. By beaming electrons onto a frozen sample of Huc that was gathered from M. smegmatis, the researchers mapped out the enzyme's atomic structure and the electrical pathways it uses to carry the electrons so that they form a current.
The team discovered that at its center, Huc has a structure, called an active site, that contains charged ions of nickel and iron. Once hydrogen molecules (made up of two protons and two electrons) enter the active site, they become trapped between the nickel and iron ions and get stripped of their electrons. The enzyme then sends these electrons along in a flowing stream to generate a current.
"The electrons are absorbed by Huc (specifically the nickel ion), and transferred to the surface of Huc (by a molecular wire formed by clusters of iron and sulphur ions)," Grinter said. "If we immobilize Huc on an electrode, the electrons can enter an electrical circuit from the enzyme surface and generate current."
Further experiments revealed that the isolated Huc enzyme can be stored for prolonged periods; that it survives being frozen or heated up to 176 degrees Fahrenheit (80 degrees Celsius); and that it can consume hydrogen at concentrations as minuscule as 0.00005% of that found in the air we breathe. These attributes, alongside the microbe's ubiquity and ability to be easily grown, could make the enzyme an ideal candidate for a power source in organic batteries, the researchers say.
"Huc can extract energy from hydrogen in the air, which is effectively limitless," Grinter said. "The amount of electricity that can be generated from the low concentrations of hydrogen in the air will be modest. This will limit the application of Huc in this context to devices that require a small but sustained amount of power. A complementary use of Huc would be in fuel cells where a higher concentration of hydrogen is provided."
Ben Turner is a U.K. based staff writer at Live Science. He covers physics and astronomy, among other topics like tech and climate change. He graduated from University College London with a degree in particle physics before training as a journalist



Reader Comments
Get you about 200,000 balloons......maneuver them towards the south pole so they get in the wind current at 10 hPa (high up).
Let them balloons (which are connected) commence to circling around at high velocity.
Going round and round and round....
I suspect there is some "free energy" in that - delivered directly from the big yellow ball in the sky.
What you think - is this a good idea or not?
Ken
Sun supplies solar and matter, we only feebly harness the solar/photon component, not the matter component.
I'll tell you this though - there is no such thing as free energy, but the amount of energy the balloons would "absorb" and send down for practical use would be a teeny teeny tiny little ittie bit of the energy there, so basically one could characterize it as "free".
How you like them apples?
Plus - tis electromagnetic energy....not from photons and but a reflection of mass.
I'm not sure photons even exist, but I know there is electricity and magnetism and they are close.
"Hydrogen is a clean fuel that, when consumed in a fuel cell, produces only water. Hydrogen can be produced from a variety of domestic resources, such as natural gas, nuclear power, biomass, and renewable power like solar and wind. These qualities make it an attractive fuel option for transportation and electricity generation applications. It can be used in cars, in houses, for portable power, and in many more applications.
Hydrogen is an energy carrier that can be used to store, move, and deliver energy produced from other sources.
Today, hydrogen fuel can be produced through several methods. The most common methods today are natural gas reforming (a thermal process), and electrolysis. Other methods include solar-driven and biological processes."
[Link]
Yes, without trying to vacuum the Hydrogen ions out of space, hydrocarbons are the most available, least expensive source for H on an industrial scale.
Sadly Hydrogen makes up only a small fraction of most hydrocarbons (3% - 7% typically) and its removal leaves a carbon-rich gas which must be condensed or partially oxidized to make 'things', like coke for steel fabrication, graphite for pencils and BBQ brickettes, etc.
According to Greta, we can no longer use the carbon-rich gas or liquid as a fuel now, can we?
Gaseous hydrocarbons contain anywhere from 1,000 Btu of energy per Ft3 (methane/nat. gas) to more than 2,000 Btu of energy per Ft3 (propanebutane). Hydrogen contains less than 250 Btu of energy per Ft3.
This is bad for two reasons; 1- You must waste 3/4 of your methane fuel (more if its propane) to get the Hydrogen out. 2- You only have 1/4 (or less) of the energy capacity available to you in the same size and pressure range of storage tank that usually holds methane.
The actual energy consumption required to get Hydrogen to market as a consumable far exceeds the energy available from the Hydrogen once it's available. Not only do you have the refining 'costs' of the original hydrocarbon but now you have the 'cost' to strip out the Hydrogen, pump the hydrogen through specially designed pipes and vessels (Hydrogen will crack normal steel and will leak past seals and valves that would normally hold a heavier gas), plus compress it multiple times between the refinery and the end users location.
It is a scam.
Hold my beer......