A chemical bond between two metal atoms has been filmed breaking and forming for the first time - something scientists say they only dreamed of seeing. Watching this happen in real time "was absolutely unbelievable", says Andrei Khlobystov at the University of Nottingham, UK, who led the team that recorded it.
"We were very excited," says Khlobystov. "We knew exactly what was happening because we recognised this as a chemical process straight away, but it was a surprise because we simply did not expect we would see it at this level of detail," he says.
Khlobystov and his team used a powerful microscope to fire a beam of electrons at a molecule composed of two atoms of the transition metal rhenium. The electron beam collided with the rhenium molecule, providing the energy for the bond between the atoms to break, while also allowing the electrons to be detected on the other side of the molecule to create an image.
As the bond broke, the atoms changed shape from circular to more elongated. Khlobystov thinks this represents a transition period between the intact and completely broken bond. During this transition, the atoms aren't entirely independent, he says. "Somehow, they feel each other."
Khlobystov says the video, and being able to see how bonds form and break, will change the way chemistry is taught at schools and universities. "It's real, we can take pictures and prove how it happens," says Khlobystov.
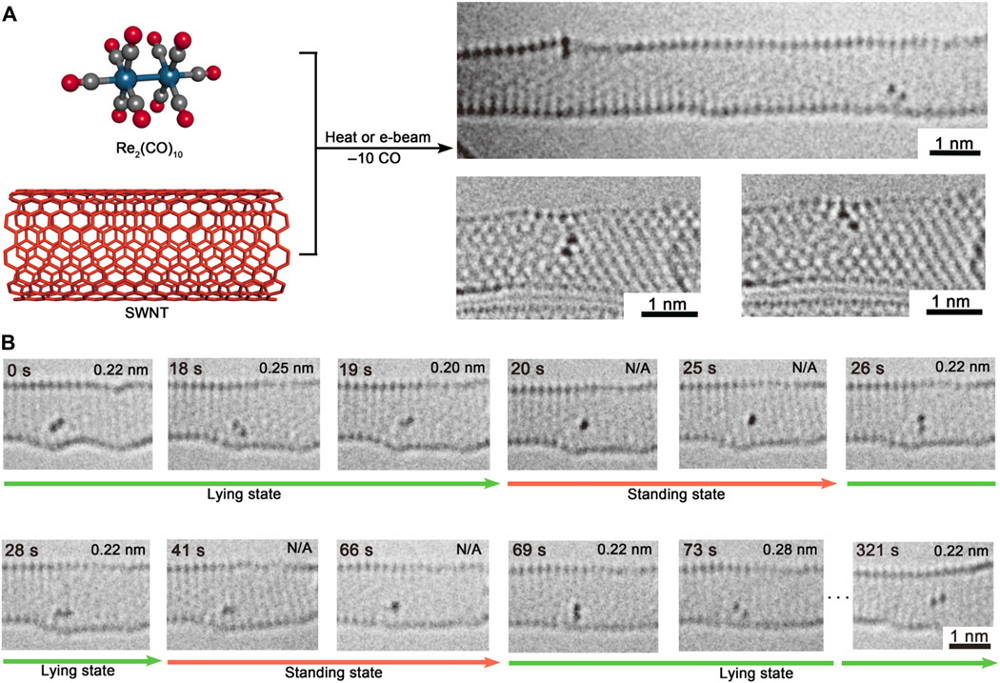
(A) Schematic illustration of the preparation of dirhenium molecule confined in SWNT by eliminating CO groups from Re2(CO)10 precursor. Three dirhenium molecules in two SWNTs can be observed in the AC-HRTEM images [two dirhenium molecules imaged at 80 kV with Cs-corrected (top image) and one dirhenium molecule Cc/Cs-corrected (bottom two images) TEM, unprocessed data]. (B) Time-series AC-HRTEM images (80 kV) from movie S1 acquired by Titan CS-corrected TEM showing the characteristic states of the dirhenium molecule changing under e−-beam irradiation. This dirhenium molecule is the right one in the SWNT of the top AC-HRTEM image in (A) named “Titan Re2 A.” The corrected Re–Re bond length of each frame is presented; error is ±0.015 nm. N/A is the standing state with unknown bond length.
"Chemical bonding is one of the most fundamental and easily grasped concepts in chemistry at a general level, but it is actually highly complex and very difficult to study in detail," says Steve Liddle at the University of Manchester, UK. "That's the beauty of this," he says. "You are as close to visualising the molecule for real as you can really ever get."
Journal reference: Science advances, DOI: 10.1126/sciadv.aay5849




BTW, how can I be sure the imagery is not simulated ? Think NASA ...