"You have to move that piece if you've touched it," my opponent says, flashing a wry grin.
Fine. I move the bishop. It's becoming increasingly obvious to me now - I'm going to lose a game of chess to a 12-year-old.
My opponent is Tanner Collins, a seventh-grade student growing up in a Pittsburgh suburb. Besides playing chess, Collins likes building with Legos. One such set, a replica of Hogwarts Castle from the Harry Potter books, is displayed on a hutch in the dining room of his parents' house. He points out to me a critical flaw in the design: The back of the castle isn't closed off. "If you turn it around," he says, "the whole side is open. That's dumb."
Though Collins is not dissimilar from many kids his age, there is something that makes him unlike most 12-year-olds in the United States, if not the world: He's missing one-sixth of his brain.
Collins was three months shy of seven years old when surgeons sliced open his skull and removed a third of his brain's right hemisphere. For two years prior, a benign tumor had been growing in the back of his brain, eventually reaching the size of a golf ball. The tumor caused a series of disruptive seizures that gave him migraines and kept him from school. Medications did little to treat the problem and made Collins drowsy. By the day of his surgery, Collins was experiencing daily seizures that were growing in severity. He would collapse and be incontinent and sometimes vomit, he says.
When neurologists told Collins' parents, Nicole and Carl, that they could excise the seizure-inducing areas of their son's brain, the couple agreed. "His neurologist wasn't able to control his seizures no matter what medication she put him on," Nicole says. "At that point, we were desperate... His quality of life was such that the benefits outweighed the risks."
Surgeons cut out the entire right occipital lobe and half of the temporal lobe of Collins' brain. Those lobes are important for processing the information that passes through our eyes' optic nerves, allowing us to see. These regions are also critical for recognizing faces and objects and attaching corresponding names. There was no way of being sure whether Collins would ever see again, recognize his parents, or even develop normally after the surgery.
And then the miraculous happened: Despite the loss of more than 15 percent of his brain, Collins turned out to be fine.
"We're looking at the entire remapping of the function of one hemisphere onto the other."The one exception is the loss of peripheral vision in his left eye. Though this means Collins will never legally be able to drive, he compensates for his blind spot by moving his head around, scanning a room to create a complete picture. "It's not like it's blurred or it's just black there. It's, like, all blended," Collins tells me when I visit him at home in January. "So, it's like a Bob Ross painting."
Today, Collins is a critical puzzle piece in an ongoing study of how the human brain can change. That's because his brain has done something remarkable: The left side has assumed all the responsibilities and tasks of his now largely missing right side.
"We're looking at the entire remapping of the function of one hemisphere onto the other," says Marlene Behrmann, a cognitive neuroscientist at Carnegie Mellon University who has been examining Collins' brain for more than five years.
What happened to Collins is a remarkable example of neuroplasticity: the ability of the brain to reorganize, create new connections, and even heal itself after injury. Neuroplasticity allows the brain to strengthen or even recreate connections between brain cells-the pathways that help us learn a foreign language, for instance, or how to ride a bike.
The fact that the brain has a malleable capacity to change itself isn't new. What's less understood is how exactly the brain does it. That's where Behrmann's study of Collins comes in. Her research question is twofold: To what extent can the remaining structures of Collins' brain take over the functions of the part of his brain that was removed? And can science describe how the brain carries out these changes, all the way down to the cellular level?
Previous neuroplasticity research has shed light on how the brain forms new neuronal connections with respect to memory, language, or learning abilities. (It's the basis for popular brain-training games meant to improve short-term memory.) But Behrmann's research is the first longitudinal study to look closely at what happens in the brain after the regions involved in visual processing are lost through surgery or damaged due to a traumatic brain injury.
"We know almost nothing about what happens in the visual system after this kind of surgery," she says. "I think of this as kind of the tip of the iceberg."
So far, Behrmann's findings are turning medical dogma on its head. They suggest that conducting brain surgeries in kids suffering seizures shouldn't be viewed as the last available option, as it was for Collins. The surgery he underwent, while successful roughly 70 percent of the time, is still uncommon, which means that many people with similar brain tumors may be suffering unnecessarily. And depending on what Behrmann discovers, we may learn more than we ever have before about the brain's capacity to bounce back.
The first time Collins collapsed because of a seizure, he was four and being minded by a babysitter. Over time, his symptoms grew more varied and more severe. "It's like my brain froze," he says. "I was really confused, and then I'd get really nauseous, throw up, and then I'd be kind of acting normal again."
A daily ritual ensued: Collins would go to school, have a seizure, collapse, and go home. Still, despite the misery, the seizures were a blessing in disguise. They led to the discovery of the tumor slowly enveloping a piece of his brain.
"These are some of the most common tumors we see in children," says Christina Patterson, MD, a pediatric epilepsy neurologist and part of the medical team that prepared Collins for surgery at the UPMC Children's Hospital of Pittsburgh. "Taking out the tumor is ultimately the cure."
The deeper problem with pediatric tumors like the one Collins developed - beyond the nausea, headaches, and confusion that he experienced - is that the seizures they produce can damage the electrical networks of the brain.
"We know that the pediatric brain has plasticity, [and] that we're constantly creating new algorithms in the brain to live life," Patterson says. "But when you have seizures on top of that, those disrupted electrical networks that are the seizures prevent any kind of meaningful remapping."
Inside our brains are about 100 billion neurons. These neurons build thousands of connections with one another and communicate with their cellular brethren by converting electrical signals into chemical neurotransmitters, which are responsible for carrying information between the brain cells. As we master new skills, the brain's neurons form new connections and strengthen old ones that aided in learning that information. Instead of discrete regions carrying out specific tasks, the brain depends on groups of neural networks talking to each other across multiple regions. (Behrmann says a single neuron can communicate with 50,000 other cells.) If the network is damaged, the brain cells can't communicate effectively.
Picture a map of the United States that shows a phone company's LTE network crisscrossing the country, and you have a rough approximation of how the human brain operates. Surgery for Collins, in this case, was akin to repairing a downed cell tower.
Before Collins' surgery to remove the tumor, doctors opened up his head and placed electrodes on the surface of his brain and inside his visual cortex. For seven days, Collins lay in a hospital bed as the electrodes mapped his brain's electrical activity, creating what was essentially a schematic diagram showing doctors where the seizures were originating and which brain areas needed to be cut out.
Collins recognized his parents after the surgery, but he couldn't match their faces to their names. The problem resolved itself in a couple of days, but the episode left Nicole and Carl concerned: How was their son's brain going to function with a missing part?
Consider, for a moment, a page from a Where's Waldo? book. When your eye focuses on the crowded image, you're actually only receiving two types of feedback: the light that falls on the retina and the color of that light. "That's all your eye can pick up," Behrmann says. "Yet somehow, almost instantaneously, you get an interpretation of the scene."
Patterson put the Collins family in touch with Behrmann, who studies how brain plasticity relates to vision at her lab at Carnegie Mellon. Collins was the ideal candidate for Behrmann's research. Children's brains are young and still developing and therefore have the most potential for neuroplastic change. Because Collins' tumor formed in the part of the brain crucial for visual processing, Behrmann could track his progress over time to determine whether there were any lingering deficits in his ability to interpret images. Because Collins was a child, his brain was also in a critical period of development where it builds the capacity to recognize faces, something that happens gradually and becomes more finely tuned throughout our teenage years.
As University of Toronto psychiatrist Norman Doidge notes in his 2007 book, The Brain That Changes Itself, the notion that there is a critical period of brain development is one of the most important discoveries in the area of neuroplasticity - and one for which we have kittens to thank. In the 1960s, as Doidge recounts, scientists David Hubel and Torsten Wiesel mapped the visual cortex of kittens - much in the same way Collins' surgical team mapped his own brain - to learn how vision is processed. Then, in an admittedly grisly procedure, the scientists sewed shut the eyelid of one of the kittens in the study. Upon opening the eyelid, they found that the visual areas of the kitten's brain responsible for processing images from that eye didn't develop, leaving the kitten blind in that eye, even though nothing was biologically wrong with the eye. The researchers discovered that if kittens' brains were to develop normally, they had to be able to see the world around them between their third and eighth weeks of life.
But another discovery from the study proved even more important - and earned Hubel and Wiesel the Nobel Prize. "The part of the kitten's brain that had been deprived of input from the shut eye did not remain idle," Doidge writes. "It had begun to process visual input from the open eye, as though the brain didn't want to waste any 'cortical real estate' and had found a way to rewire itself."
In Collins' case, the question was whether the fully intact left hemisphere of his brain would pick up the functionality of the missing third of his brain, especially the task of facial recognition, which is typically carried out by the right hemisphere.
Collins' left brain not only looked and performed the way his left brain should; it also looked similar in scans to other kids' intact right brains.Starting just before Collins was seven and continuing for three years, Behrmann administered a series of tests roughly every six months. In one challenge, he was shown photos of faces in intervals of roughly 30 seconds. If he remembered a face, he clicked a button. A similar test was administered using photos of houses, and if Collins saw the same photo back to back, he clicked a button. Each test occurred while he was inside a functional MRI machine, which allowed Behrmann to measure the flow of blood and oxygen to different regions of the brain. The more active an area of the brain, the more blood it draws.
Throughout these experiments, Behrmann compared Collins' brain function to a control group of kids his own age without brain abnormalities. The results, published last August in Cell Reports, were striking: His neurological function was "absolutely normal," with no subtle delays or deviations in development.
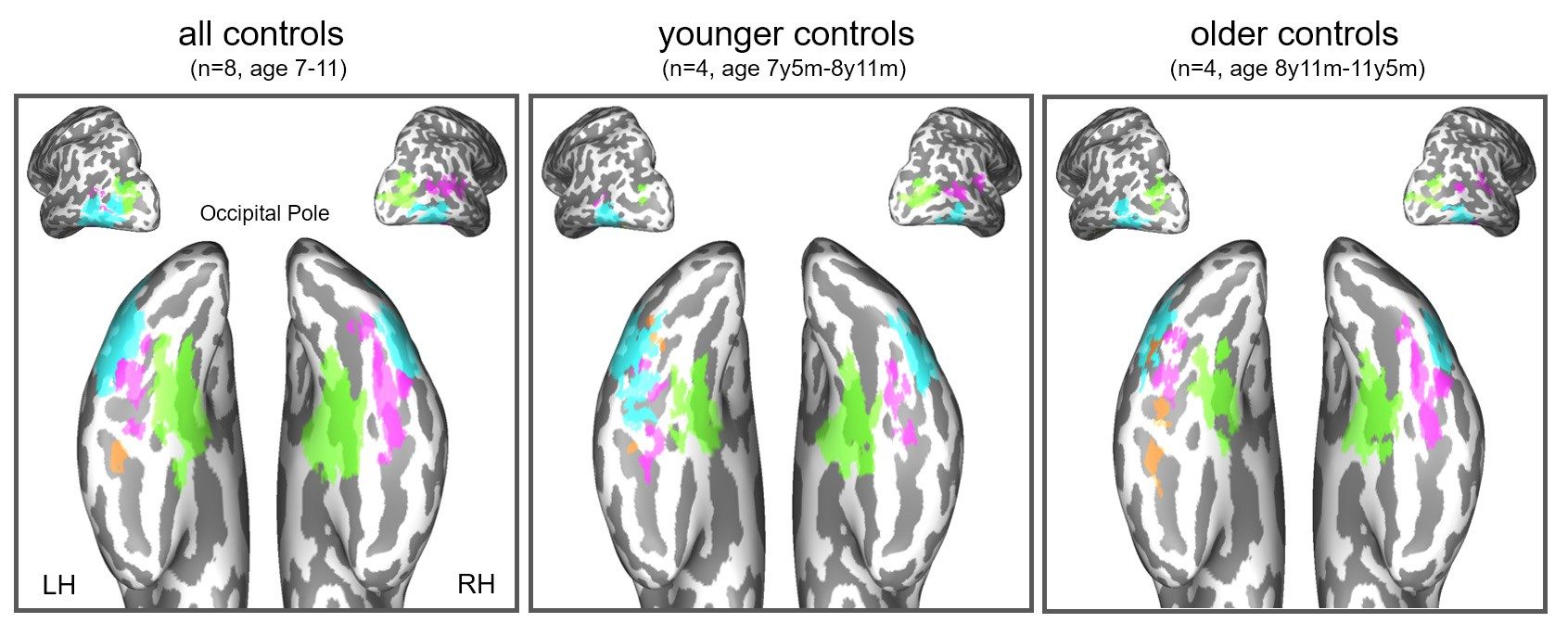
In scans, Collins' left brain not only looked and performed the way his left brain should; it also looked similar in scans as other kids' intact right brains. That's because the functions of the visual cortex he lost by having one-third of his right brain removed - the ability to see objects and know what they are, and the ability to recognize faces - were subsumed by his left brain. Also fascinating to Behrmann was how the left brain could accommodate two different skills: word recognition, which is the domain of the left brain, as well as facial recognition. Indeed, part of the surprise was that the left brain could keep doing what it normally does in addition to the newly added right-brain activity.
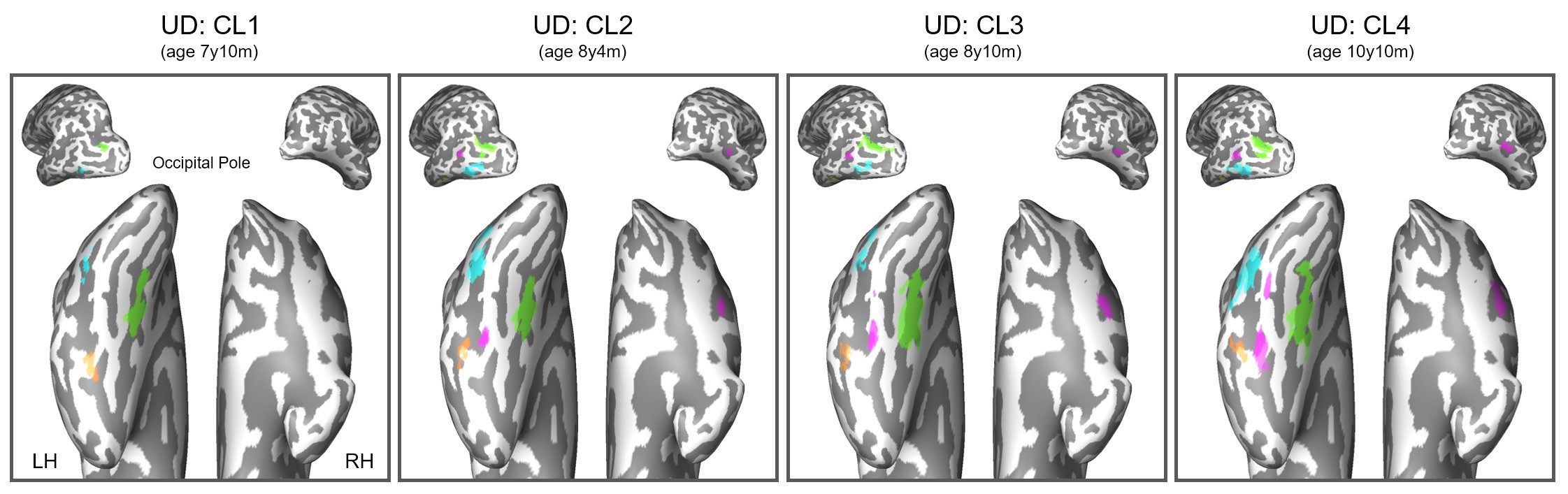
Just how the brain accomplishes this feat remains a central question. By analyzing brain scans using a neuroimaging technique known as diffusion tensor imaging, which shows how water travels along the brain's white-matter tracts, Behrmann has found initial glimmerings that the white matter of the brain - the electrical wiring that underlines communication between multiple neurological regions - actually changes. Areas of the brain that weren't connected before create new links, an example of neuroplasticity in action that may preserve brain functionality. But scientists still don't know what triggers the cells of the white matter to behave in this way.
"When Tanner is 20, I think we'll know a lot more about the overall wiring," Behrmann says. "The one thing that we will not know in humans, and I don't know how we will ever know it, are the changes that occur at the level of the cells themselves."
Every three to six months, Collins returns to Behrmann's lab to undergo tests and be examined for any visual deficits. Behrmann hopes that following him over time will lead to more definitive answers, not only about how his visual system finally reorganizes itself but also the process by which it does so. "We've got a long way to go, but the work, I think, is really exciting," she says.
In a follow-up study Behrmann conducted with Collins and nine other children - all of whom are missing areas of either their left or right hemisphere - eight of them, including Collins, showed absolutely normal vision function. The two who did not are children whose brain damage from seizures was more severe prior to their surgeries.
This sort of insight is needed to gauge when to perform a brain surgery like the one Collins had. At what age should parents agree to remove a tumor that's causing epileptic seizures? Sometimes, resective surgery that removes brain tissue can make it difficult for a person to use and understand words; it can also, as it did in Collins' case, result in visual impairment.
"Once we have a better picture of exactly what happens after we remove large segments of the brain, we may be able to counsel families more effectively," says Taylor Abel, MD, a pediatric neurosurgeon who specializes in epilepsy surgery and arrived at the Children's Hospital of Pittsburgh last summer to begin collaborating with Behrmann. "The goal should be to do whatever you can to stop the seizures and get off of medications as early in your life as possible. The sooner you do that, the sooner you can return to a normal developmental trajectory."
It may even be the case, Abel and Behrmann point out, that some of the reorganization that took place in Collins' brain started prior to his scheduled surgery. It's not something Behrmann can prove, since all the research conducted on Collins has taken place post-surgery.
"When you have an abnormality in your brain that's causing seizures, that abnormality can actually cause the brain to reorganize or start reorganizing before the surgery actually takes place," Abel says. "But the other thing that sometimes happens is that the seizures affect the functions in the brain, and the brain doesn't reorganize."
Behrmann says one of the fundamental goals of her research is to study a large enough population of children to determine if there are patterns of optimal recovery based on the age they had their surgery. Reorganization to the degree Collins has experienced is impossible for adults undergoing similar surgery, Behrmann says, as they lack the neuroplasticity seen in children.
For Nicole and Carl, the surgery was unequivocally the right decision. "What was happening before the surgery was pretty awful," Nicole says. "After surgery, the changes were only for the better. Yeah, he has his visual deficits. But everything else was for the better."
In late 2017, a follow-up MRI at the Children's Hospital of Pittsburgh showed that Collins' tumor grew back. This time, though, it was the size of a pea. Two months later, in February 2018, surgeons opened his brain a second time. Collins says the prospect of a second surgery didn't bother him; he just wanted the pea-size tumor out of his head so he wouldn't have to worry about it. (The surgery went well, and he's still tumor-free.)
As we close in on minute 24 of our chess match, I move my king in the corner of the board, still certain of my impending doom. Collins scans his remaining white pieces and then takes a look at where his king sits.
"Mate," he says, looking up at me.
Checkmate for me, I realize, surprised by a victory I did not expect. Collins begins breaking down the moves he made, retracing some of his steps. It seems he forgot about a pawn of mine that was still on the board.
"I like losing," he says. "Obviously, I like winning, too. But when you lose, you gain the knowledge."
Even after losing a portion of his brain, Collins is still learning. His brain is still growing, still adapting - and, even if it's not readily apparent, still changing.




Reader Comments
A boy is whole despite losing 1/6th of his brain. Must be 'soul' that makes him who he is... according to the ID theory.
But Phineas Gage loses a small part of his frontal medial cortex, turns into a hustler psycho- that's ignored.
The brain has parts that are singular and cannot be easily retrained. In this boy's case he was lucky those parts were still in tact.
Watch the series by David Eagleman about the brain that was on PBS, and you see a case of a girl with epilepsy who had one half of her brain removed to stop seizures and she too is doing well.
But if you take a spike and put it in their frontal cortex, like Gage got, you have to question what exactly is a soul if empathy is so easily wiped out that way.