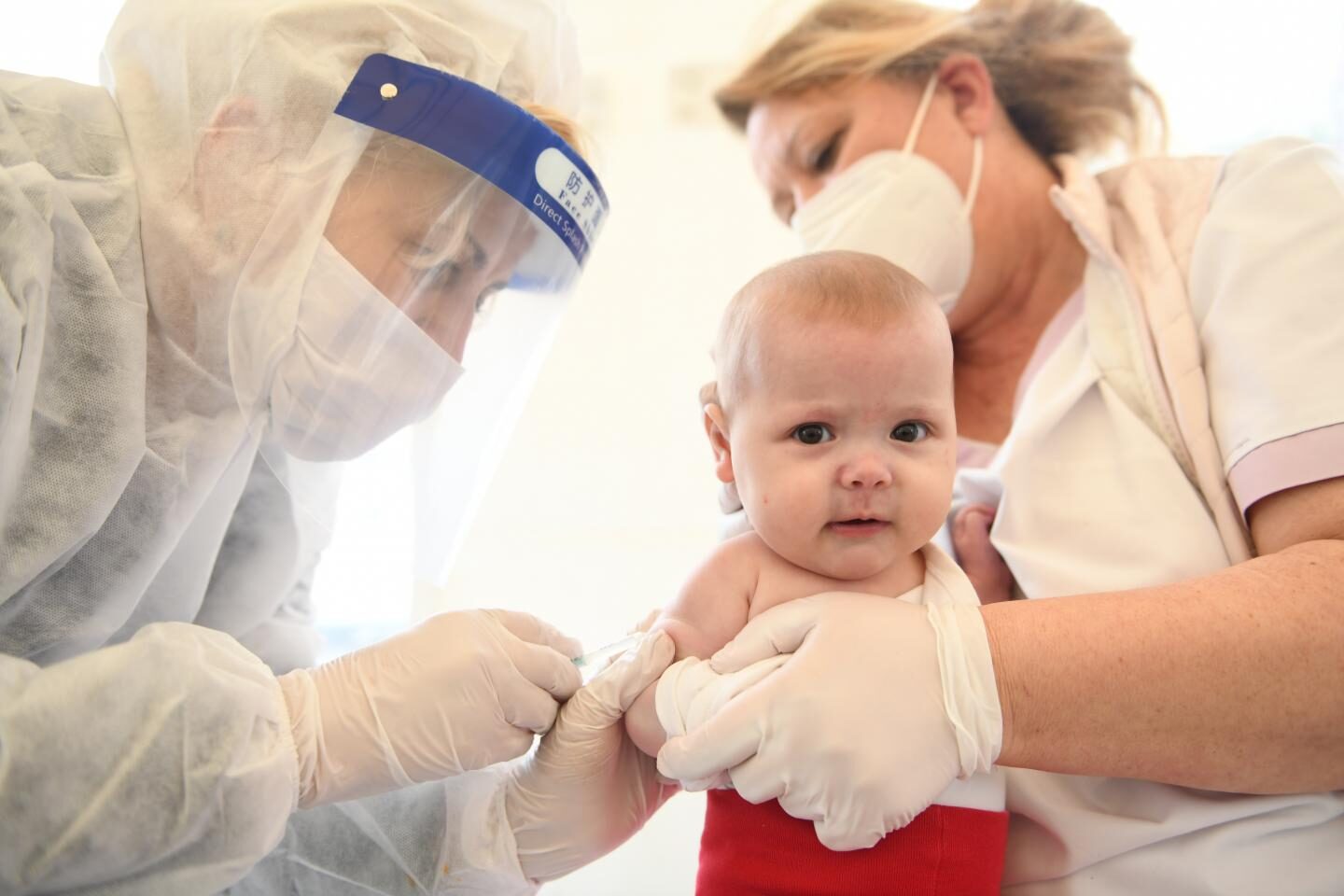
The Bloc's drug watchdog gave the green light for children older than six months to get either Pfizer or Moderna's jab.
It comes amid growing fears of another Covid wave this winter.
British authorities have so far held out on approving jabs for infants despite massive pressure, due to concerns that the benefits don't outweigh any potential risks.
Children rarely get seriously ill with the coronavirus, and the majority are thought to have already been infected.
The European Medicines Agency, which signed off on the move today, said that the doses would be 'lower' for infants.
Children in the age bracket whose parents want them to be jabbed with Pfizer's will be offered a dose of 3micrograms.
For comparison, adults in Britain get a dose 10 times stronger. Older children, who are allowed to be vaccinated in the UK, get given a dose of up to 10micrograms.
Slightly stronger doses are recommended by the EMA for children given Moderna's vaccine (25micrograms).
This is half the dose given to older children in Britain, and a quarter of that approved for adults.
Lower doses are given to children to avoid complications, such as myocarditis — the rare condition that spooked health chiefs early on in the pandemic.
The EMA's recommendations, which have already sparked concerns, will be sent to the European Commission for a final decision.
Scores of countries, including the US, China and Israel, already offer vaccines to kids under five, despite huge backlash over the moves.
Professor Paul Hunter, an epidemiologist based at the University of East Anglia, told MailOnline: 'The issue is whether or not they (young children) actually need it.'
Some children in at-risk groups could benefit from a vaccine but virtually all children have had one or more Covid infections by now, he said.
'I do not see value in vaccinating children that young unless they are susceptible to severe disease,' Professor Hunter said.
He noted that medical agencies may rule a vaccine is safe to use in an age group 'but does not necessarily mean it would be used in that context'.
Professor Hunter added: 'Just because it can be used do not mean it will be used.
Comment: And what if vaccine mandates come back in?
'I think we are at a stage of the pandemic where if you have had a number of infections, your risk of severe outcomes is lower.
'Some children may benefit but I would not think this is many.
Comment: That's highly doubtful.
'For example, in the 12 to 15 age group in the UK, only half have taken up a vaccine. So I think uptake in younger groups would not exceed this.
'I would be very surprised if the UK's vaccine committee advised that the vaccine should be used in young children unless there are unusual circumstances.'
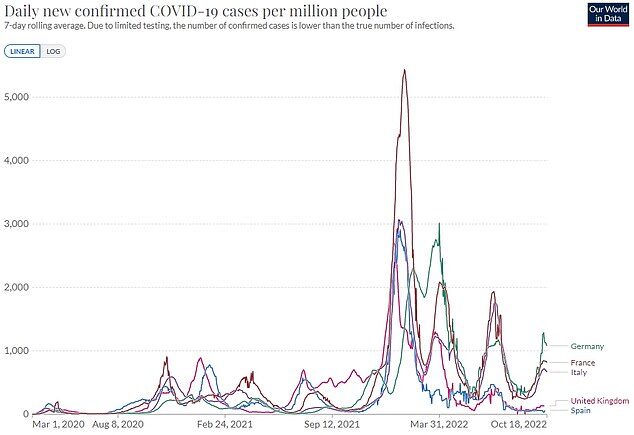
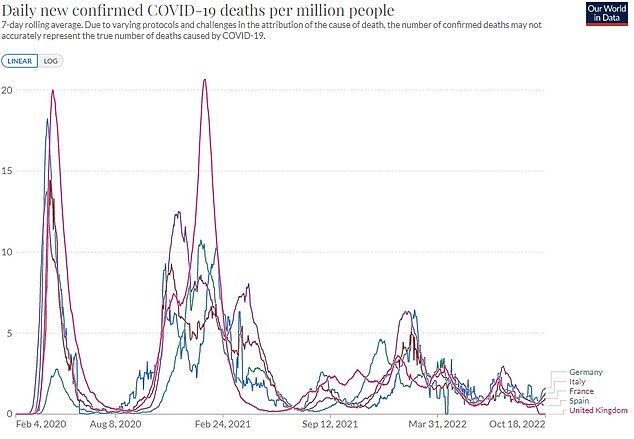
Despite fears of winter chaos, infection rates are already beginning to slow down in EU nations.
Outbreaks appear to have stalled in Germany, France and Italy, but data is much less accurate because fewer tests are being carried out.
Comment: So outbreaks have stalled but they are approving vaccines for babies... how does that work?
Cases are also on the up in Britain, latest data shows.
Health chiefs have already started to crank up their Covid advice in response to the trend, urging unwell Britons not to see elderly relatives.
Hospitals themselves have already reimposed mask rules for visitors and patients.
Others have brought back social distancing guidelines, in scenes reminiscent of the darkest days of the pandemic.



Comment: #PfizerLiedPeopleDied: