The FDA's vaccines committee met on Friday to discuss the approval of booster shots of Pfizer's Covid-19 vaccine for individuals 16 years and older. After a series of presentations from the Centers for Disease Control and Prevention (CDC), academics, and Pfizer representatives, the FDA's 15-member panel will cast their votes on Friday afternoon.
In August, FDA gave Pfizer's existing two-shot regimen its approval, paving the way for the Biden administration to unveil a program of tough new mandates.
Meanwhile, case numbers of Covid-19 are rising throughout the world, even in the most widely vaccinated countries. Prior to the Friday meeting, Pfizer disclosed the results of several real-world studies that all showed the company's two-shot dosing losing its effectiveness against the virus in the weeks and months after injection.
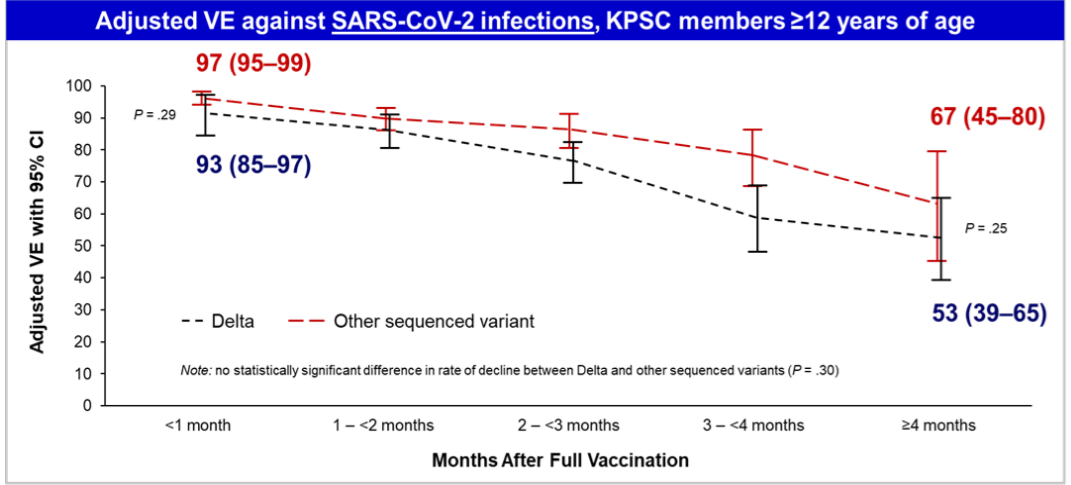
Data from Israel, which is currently rolling out booster shots to most of its population, shows that two months after their second jab, the risk of infection more than doubles. Another Israeli study found that, by summer, those vaccinated in January with Pfizer's shot were only 39% protected against infection and 40% protected against symptomatic infection, down from 95% at the time of vaccination. Two studies published last month by the CDC showed "similar reductions in effectiveness," Pfizer noted.
Pfizer recommended that booster shots be rolled out before the fall/winter flu season, a recommendation shared by the Biden administration, which announced last month - before the FDA even hinted at approval - that boosters would be given out by the end of September. White House coronavirus adviser Dr. Anthony Fauci also stated at the beginning of the month that a third dose would "likely" be needed for Americans to qualify as "fully vaccinated."
Biden's booster plans caused grumblings within the FDA. Prior to Friday's vote, two high-level FDA vaccine officials unexpectedly resigned, while the agency itself expressed skepticism over the real-world data provided by Pfizer. "There are known and unknown biases" that can affect the reliability of such observational studies, they said, stating that "currently US-licensed or authorized Covid-19 vaccines still afford protection against severe Covid-19 disease and death."
Pfizer's shot is the only coronavirus vaccine approved by the FDA, and the only one approved for use on under-18s. Vaccines by Moderna and Johnson & Johnson have received emergency-use authorizations.
As Pfizer prepared to make its case for boosters with the FDA, Moderna released a study on Wednesday showing that its vaccine also loses efficacy with time. "The increased risk of breakthrough in this analysis quantifies the impact of waning immunity...," the pharma firm said. "The company believes this adds to evidence of potential benefit of a booster dose."




Comment: