OF THE
TIMES
Maybe the militaries will shut down their sonar in the Subs before they kill all sea life.
Trump card is the court joker...aka clown world. period... case closed: diagnosis "unfit to lead" the U.S. is the laughing stock of the world....
Send the Nazi Zionist back to the Ukraine and put him on the front lines. Putin will show him what he thinks of child rapers. :O
... and just when things look their darkest... Bug-eyed humanoids from Alpha Centauri! They love the taste of humans - well, if the humans have...
Unlikely allies, forced together, to try and keep the Axis of evil, at bay. The schoolyard bullies are now having to think twice before committing...
To submit an article for publication, see our Submission Guidelines
Reader comments do not necessarily reflect the views of the volunteers, editors, and directors of SOTT.net or the Quantum Future Group.
Some icons on this site were created by: Afterglow, Aha-Soft, AntialiasFactory, artdesigner.lv, Artura, DailyOverview, Everaldo, GraphicsFuel, IconFactory, Iconka, IconShock, Icons-Land, i-love-icons, KDE-look.org, Klukeart, mugenb16, Map Icons Collection, PetshopBoxStudio, VisualPharm, wbeiruti, WebIconset
Powered by PikaJS 🐁 and In·Site
Original content © 2002-2024 by Sott.net/Signs of the Times. See: FAIR USE NOTICE
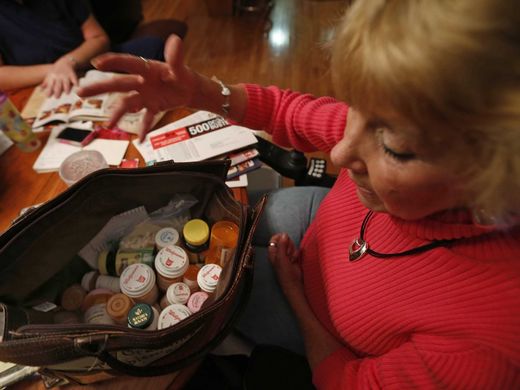
Comment: For more on hepatitis C and its treatment listen to the latest episode of Sott's Health and Wellness Show.