"I remember thinking to myself that I would bet anything that [it] didn't work that way," biochemist Leslie Orgel wrote of his meeting with Mitchell half a century ago. "Not since Darwin and Wallace has biology come up with an idea as counter-intuitive as those of, say, Einstein, Heisenberg and Schrödinger."
Over the following decades, however, it became clear that Mitchell was right. His vindication was complete when he won a Nobel prize in 1978. Even today, though, most biologists have yet to grasp the full implications of his revolutionary ideas - especially for the origin of life.
"Mitchell's ideas were about how cells are organised in space, and cellular energy generation is a feature of that," says geochemist Mike Russell of NASA's Jet Propulsion Laboratory in Pasadena, California. "The problem is that most ideas on the origin of life lack both spatial organisation and a supply of energy to drive replication or growth."
A few researchers, including Russell, have been rethinking the origin of life in the light of Mitchell's ideas. They think the most counter-intuitive trait of life is one of the best clues to its origin. As a result, they have come up with a radically different picture of what the earliest life was like and where it evolved. It's a picture for which there is growing evidence.
Before Mitchell, everyone assumed that cells got their energy using straightforward chemistry. The universal energy currency of life is a molecule called ATP. Split it and energy is released. ATP powers most of the energy-demanding processes in cells, from building proteins to making muscles move. ATP, in turn, was thought to be generated from food by a series of standard chemical reactions. Mitchell thought otherwise. Life, he argued, is powered not by the kind of chemistry that goes on in a test tube but by a kind of electricity.

Mitchell dubbed his theory chemiosmosis, and it is not surprising that biologists found it hard to accept. Why would life generate energy in such a complicated and roundabout way, when simple chemical reactions would suffice? It just didn't make sense.
It might be counter-intuitive, but chemiosmosis has turned out to be ubiquitous in the living world. Proton power drives not only cell respiration, but photosynthesis too: energy from the sun is converted into a proton gradient in essentially the same way as the energy of food.
And proton gradients are often harnessed directly, rather than being used to make ATP. They drive the rotation of the bacterial flagellum, as well as the active transport of numerous substances in and out of cells. So proton power is central to energy generation, movement and maintaining the internal environment - some of the most basic features of life.
This suggests that proton power is no late innovation but evolved early in the history of life, an idea supported by the tree of life. The first branch in the tree is between the two great groups of simple cells, bacteria and archaea. Both of these groups have proton pumps and both generate ATP from proton currents, using a similar protein. The obvious explanation is that both inherited this machinery from a common ancestor - the progenitor of all life on Earth.
Think about the properties of that common ancestor, however, says Bill Martin of the University of Düsseldorf in Germany, and you come up with a very strange beast indeed. He starts from the assumption that traits found in both the archaea and bacteria are most likely inherited from the common ancestor of all life - though a few have clearly been acquired later by gene exchange. Traits that are distinct presumably evolved independently.
There is no doubt that the common ancestor possessed DNA, RNA and proteins, a universal genetic code, ribosomes (the protein-building factories), ATP and a proton-powered enzyme for making ATP. The detailed mechanisms for reading off DNA and converting genes into proteins were also in place. In short, then, the last common ancestor of all life looks pretty much like a modern cell.
Yet the differences are startling. In particular, the detailed mechanics of DNA replication would have been quite different. It looks as if DNA replication evolved independently in bacteria and archaea, according to Eugene Koonin at the National Center for Biotechnology Information in Bethesda, Maryland.
Beyond that, many biochemical pathways are catalysed by quite different enzymes. The most surprising and most significant of these is fermentation, the production of energy from food without oxygen. Fermentation is often assumed to be the primordial method of energy generation. Yet Martin has shown that the enzymes responsible are totally unrelated in archaea and bacteria. It looks as if fermentation evolved twice later on, rather than at the dawn of life.
Baffling boundaries
Even more baffling, says Martin, neither the cell membranes nor the cell walls have any details in common. "At face value, the defining boundaries of cells evolved independently in bacteria and archaea," he says.
But if that's the case, what sort of a cell was this common ancestor? A cell with no boundary? Impossible! Something unique? If you exclude the impossible, then whatever you are left with must be true.
If Martin is right, the last common ancestor of life on Earth was a sophisticated entity in terms of its genes and proteins, and was powered by proton currents rather than fermentation. Yet at the same time, its bounding membranes were apparently different to anything found today. It was life, but not as we know it.
Then, around 2002, Martin came across the work of Russell. Until that time, Russell had been a lone voice. His geochemical ideas about the origin of life didn't go down well with the molecular biologists who dominated the field.
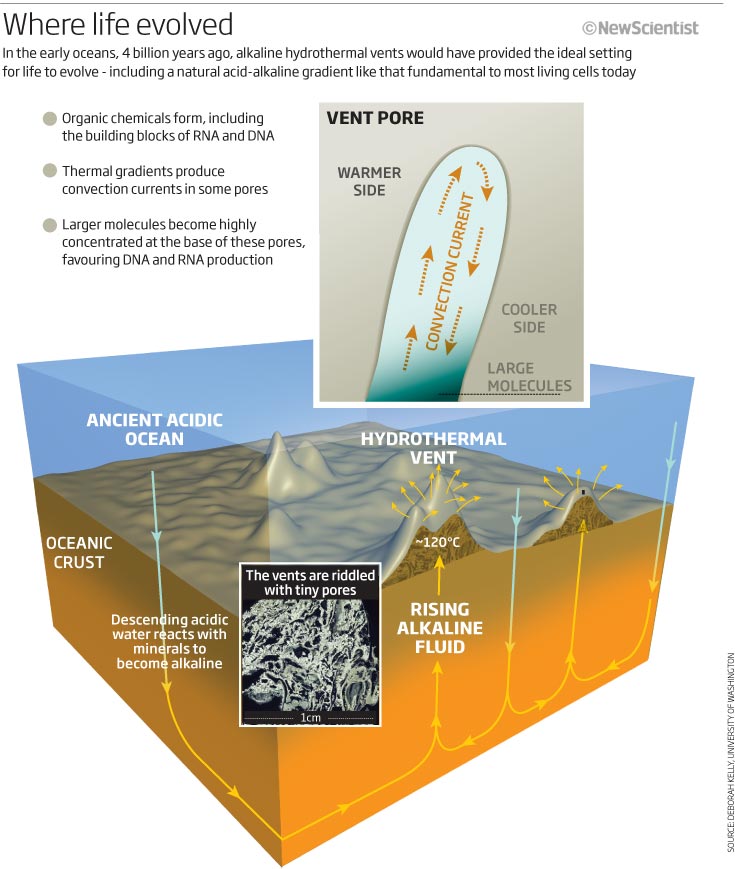
From the early 1990s, Russell had been exploring the possibilities of a very particular kind of hydrothermal vent called an alkaline vent, at the time known only from remnants found in ancient rocks. Unlike the black smokers discovered in 1977, formed by the violent reaction of seawater with volcanic lava rising up at the mid-ocean ridges, Russell's vents were much tamer affairs, little more than bubbly rocks riddled with labyrinthine pores.
These vents form when water reacts with the mineral olivine, which is common in the sea floor - and would have been even more common early on, before the Earth's crust thickened. The process produces a new mineral, serpentine, and releases hydrogen, alkaline fluids and heat. It also makes the rocks expand and crack, allowing more water to percolate down, sustaining the reaction. The warm, hydrogen-rich effluent ultimately breaks through the sea floor as an alkaline hydrothermal vent.
Interest in alkaline vents rose in 2000, when Deborah Kelley and her colleagues from the University of Washington in Seattle stumbled (if one can stumble in a submersible) across an active alkaline vent field just off the mid-Atlantic ridge, exactly where Russell said such vents should be. The team dubbed it the Lost City, partly for its spectacular spires of rock, which form as carbonates precipitate out in the alkaline fluid.
Like ancient vents, the spires of the Lost City are riddled with tiny pores, some with dimensions not dissimilar to modern cells. And the chemistry fits the bill too. A report last year confirmed the presence of methane and other small hydrocarbons, as well as hydrogen itself (Science, vol 319, p 604).
The vents themselves may be much the same as those around 4 billion years ago, but back then the oceans were very different. The primordial oceans were saturated with carbon dioxide, making them acidic, whereas the seas today are slightly alkaline. And there was practically no oxygen. Without oxygen, iron dissolves readily: the vast banded-iron formations around the world reveal just how much iron was once dissolved in oceans. As oxygen levels slowly rose, billions of tonnes of iron precipitated out as rust.
What this means, says Russell, is that the interface between the alkaline vents and the ancient seas would have been much more conducive to primordial biochemistry than they are today. In particular, bubbles of iron-sulphur minerals - which have remarkable catalytic properties - would have formed in the pores. This is not conjecture. Russell has found ancient vents with a similar structure and even reproduced them in the lab.
The fact that alkaline vents would have had a labyrinth of naturally forming microcompartments is what attracted the attention of Martin. Such compartments could have been the precursors of biological cell walls that he sought, providing a scaffold within which the stuff of cells could form. Together, Martin and Russell have pointed out that identical iron-sulphur minerals can still be found at the heart of proteins that convert carbon dioxide to sugars - using hydrogen gas - in archaea and bacteria such as methanogens and acetogens.
The vent fluid would also have contained nitrogen compounds such as ammonia, and conditions would have favoured the production of amino acids - the building blocks of proteins.
That's not all. In the presence of phosphate, minerals might have catalysed the production of nucleotides - the building blocks of RNA and DNA. And if nucleotides did form by mineral catalysis, the pores in alkaline vents would have had an extraordinary effect.
Simulations by Dieter Braun's team at the Ludwig Maximillian University in Munich, Germany, show that the temperature gradient between the top and bottom of the pores concentrates nucleotides at one end, which encourages the molecules to join together to form strings of RNA and DNA (see diagram). These larger molecules would then become concentrated to even higher levels. What's more, the convection currents would produce a continual rise and fall in temperature - as is now used to make DNA in labs across the world.
RNA world
Laboratory experiments by a team led by Nobel prizewinner Jack Szostak of Harvard University, published earlier this year, have confirmed that these conditions do indeed concentrate nucleotides and nucleic acids. The team also found that fatty acids become concentrated, leading to the spontaneous formation of cell-like bubbles inside the pores.
It's hard to imagine a better setting for the RNA world widely thought to bridge the gap between simple organic chemicals and the complexities of DNA and proteins. So the idea that ancient alkaline hydrothermal vents were the incubators for life looks very plausible even before you consider their most striking feature: a ready-made proton gradient.
Back then the seas were acidic, and acidity is defined in terms of protons - acids are rich in them. "Alkaline fluids bubbling into an acidic ocean form catalytic mineral 'cells' with a proton gradient across their inorganic membranes," says Russell. "They're set up in the same peculiar way as all cells today."
Some researchers have dismissed Russell's naturally chemiosmotic "cells" as a mere curiosity, irrelevant to the origin of life. But when Martin and Russell considered the bioenergetics of the simplest prokaryotes, they realised that the first cells could never have escaped the vents without first mastering chemiosmosis - explaining why Mitchell's bizarre mechanism is so central to life today, and so universal.
The great escape
For all the astonishing wealth of life on Earth, there are only five ways that carbon dioxide is captured and converted into living matter - and only one of those costs nothing at all. That's the straight reaction of hydrogen with carbon dioxide. This exothermic reaction converts carbon dioxide into simple organic molecules and also releases energy. It's been described as "a free lunch you're paid to eat". And while hydrogen doesn't usually bubble obligingly out of the ground, it does in alkaline vents.
The reaction of hydrogen with carbon dioxide is central to life in the vents, but there is a big problem: it costs some energy to kick-start the reaction in the first place, while the amount of energy released to fuel growth is paltry. In fact, so paltry, according to Rolf Thauer at the Max Planck Institute for Terrestrial Microbiology in Marburg, Germany, that it's impossible for such bacteria to grow by chemistry alone: they need the proton power of chemiosmosis.
To understand this, think of the energy stored by ATP as equivalent to £10. If it takes £10 to kick-start a reaction, which then releases £15, in theory a cell has gained £5. However, if the only way a cell has to store energy is to make ATP, it can make only one molecule; to make two new ATPs would cost £20. So one ATP would have been spent to gain one ATP, and the spare £5 wasted as heat. That's not consistent with being alive.
For proto-life in the vents, this would not have been an issue. The fluid from the vents would have contained reactive molecules such as methyl sulphide, which would generate acetyl phosphate, a molecule that some bacteria today still use interchangeably with ATP. What's more, the natural proton gradient would have supplemented this energy source by spontaneously generating another primitive form of ATP called pyrophosphate.
Pyrophosphate also acts in much the same way as ATP and is still used alongside ATP by many bacteria and archaea. These bacteria speed up its production using a simple enzyme called pyrophosphatase. In soon-to-be published work done with Wolfgang Nitschke at the Institute of Structural Biology and Microbiology in Marseille, France, Russell has shown that this enzyme is found in some of the most primitive cells known and crops up on both sides of the bacteria-archaea divide. That suggests it dates right back to the beginning.
So the common ancestor of life could harness the natural proton gradient of ancient vents to produce energy. To escape the vents, just one further step was necessary: reversing the process to store energy.
Chemiosmosis allows cells to save up small amounts of energy - the £5s that would otherwise be wasted. A reaction can be repeated time and time again, just to pump a proton over a membrane. Like saving up to buy something, eventually the proton gradient will be enough to produce one pyrophosphate or one ATP. The upshot is that proton gradients enable cells to grow, to leave the vents.
Indeed, it appears Mitchell's oddity is a necessary precondition for life. While breaking down sugars can provide enough energy for growth without any need for chemiosmosis, the process - glycolysis - involves complex pathways and requires lots of sugar, and is thus very unlikely to have been the main source of energy for the first life.
The picture painted by Russell and Martin is striking indeed. The last common ancestor of all life was not a free-living cell at all, but a porous rock riddled with bubbly iron-sulphur membranes that catalysed primordial biochemical reactions. Powered by hydrogen and proton gradients, this natural flow reactor filled up with organic chemicals, giving rise to proto-life that eventually broke out as the first living cells - not once but twice, giving rise to the bacteria and the archaea.
Many details have yet to be filled in, and it may never be possible to prove beyond any doubt that life evolved by this mechanism. The evidence, however, is growing. This scenario matches the known properties of all life on Earth, is energetically plausible - and returns Mitchell's great theory to its rightful place at the very centre of biology.
Read more: How it might have happened: 10 steps to the first cells



Segment
"vents themselves may be much the same as those around 4 billion years ago, but back then the oceans were very different. (...) saturated with carbon dioxide, making them acidic, whereas the seas today are slightly alkaline. And there was practically no oxygen"
needs update, especially considering it's been proven since 2001 that as far as 4.4 billion years ago Earth had oceans and an atmosphere rich oxygen. For more details, check links below:
www.nsf.gov/od/lpa/news/press/01/pr0102.htm,
[Link] Does that affect the theory?