"These are the first bacteria found to use manganese as their source of fuel," says Jared Leadbetter, professor of environmental microbiology at Caltech who, in collaboration with postdoctoral scholar Hang Yu, describes the findings in the July 16 issue of the journal Nature. "A wonderful aspect of microbes in nature is that they can metabolize seemingly unlikely materials, like metals, yielding energy useful to the cell."
The study also reveals that the bacteria can use manganese to convert carbon dioxide into biomass, a process called chemosynthesis. Previously, researchers knew of bacteria and fungi that could oxidize manganese, or strip it of electrons, but they had only speculated that yet-to-be-identified microbes might be able to harness the process to drive growth.
Leadbetter found the bacteria serendipitously after performing unrelated experiments using a light, chalk-like form of manganese. He had left a glass jar soiled with the substance to soak in tap water in his Caltech office sink before departing for several months to work off campus. When he returned, the jar was coated with a dark material.
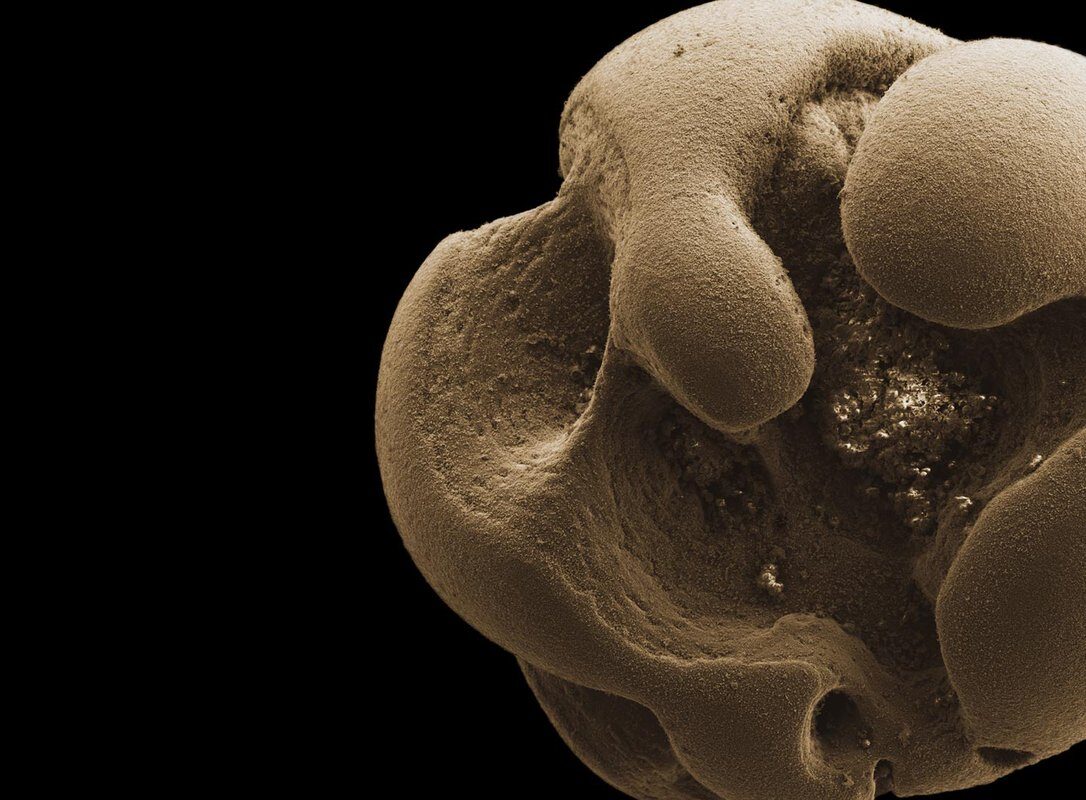
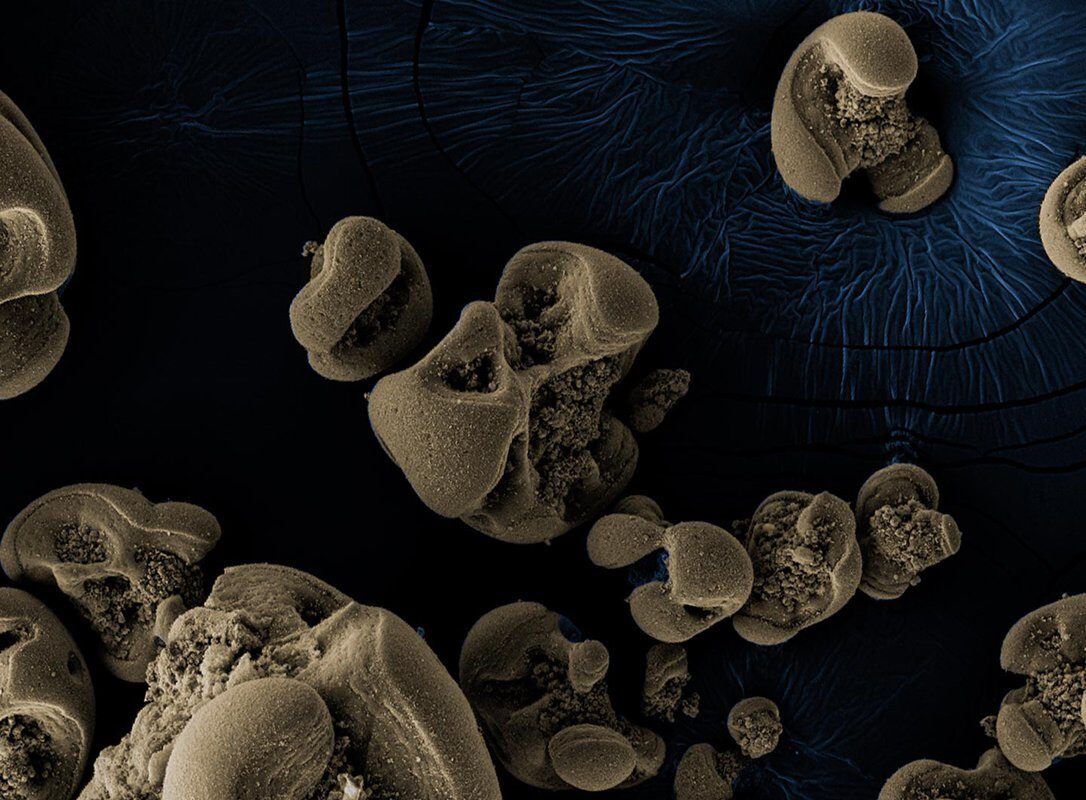
The black coating was in fact oxidized manganese generated by newfound bacteria that had likely come from the tap water itself. "There is evidence that relatives of these creatures reside in groundwater, and a portion of Pasadena's drinking water is pumped from local aquifers," he says.
Manganese is one of the most abundant elements on the surface of the earth. Manganese oxides take the form of a dark, clumpy substance and are common in nature; they have been found in subsurface deposits and can also form in water-distribution systems.
"There is a whole set of environmental engineering literature on drinking-water-distribution systems getting clogged by manganese oxides," says Leadbetter. "But how and for what reason such material is generated there has remained an enigma. Clearly, many scientists have considered that bacteria using manganese for energy might be responsible, but evidence supporting this idea was not available until now."
The finding helps researchers better understand the geochemistry of groundwater. It is known that bacteria can degrade pollutants in groundwater, a process called bioremediation. When doing this, several key organisms will "reduce" manganese oxide, which means they donate electrons to it, in a manner similar to how humans use oxygen in the air. Scientists have wondered where the manganese oxide comes from in the first place.
"The bacteria we have discovered can produce it, thus they enjoy a lifestyle that also serves to supply the other microbes with what they need to perform reactions that we consider to be beneficial and desirable," says Leadbetter.
The research findings also have possible relevance to understanding manganese nodules that dot much of the seafloor. These round metallic balls, which can be as large as grapefruit, were known to marine researchers as early as the cruises of the HMS Challenger in the 1870s. Since then, such nodules have been found to line the bottom of many of Earth's oceans. In recent years, mining companies have been making plans to harvest and exploit these nodules, because rare metals are often found concentrated within them.
But little is understood about how the nodules form in the first place. Yu and Leadbetter now wonder if microbes similar to what they have found in freshwater might play a role and they plan to further investigate the mystery. "This underscores the need to better understand marine manganese nodules before they are decimated by mining," says Yu.
"This discovery from Jared and Hang fills a major intellectual gap in our understanding of Earth's elemental cycles, and adds to the diverse ways in which manganese, an abstruse but common transition metal, has shaped the evolution of life on our planet," says Woodward Fischer, professor of geobiology at Caltech, who was not involved with the study.
The study, titled, "Bacterial chemolithoautotrophy via manganese oxidation," was funded by NASA and Caltech.



R.C.