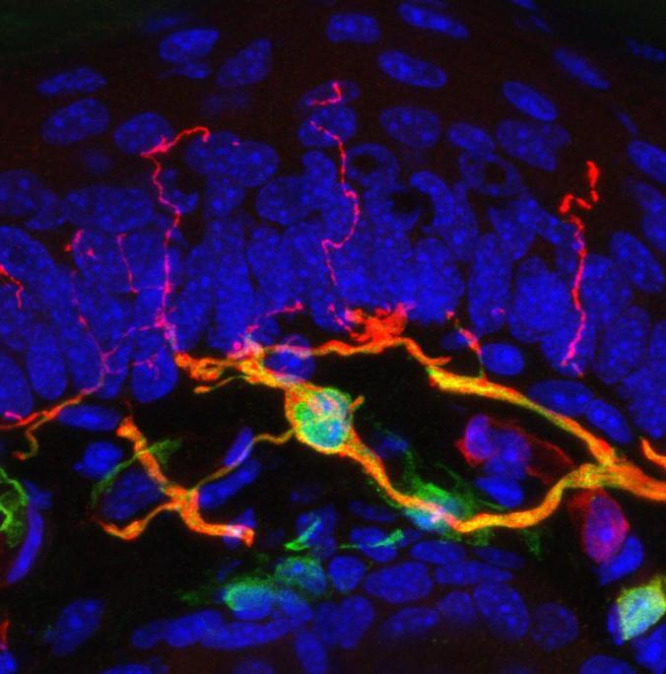
Most people who've been jabbed by a needle know the drill: First the pierce, then the sharp, searing pain and an urge to pull away, or at least wince. While the exact circuitry behind this nearly universal reaction is not fully understood, scientists may have just found an important piece of the puzzle: a previously unknown sensory organ inside the skin.
Dubbed the nociceptive glio-neural complex, this structure is not quite like the typical picture of a complex organ like the heart or the spleen. Instead, it's a simple organ made up of a network of cells called glial cells, which are already known to surround and support the body's nerve cells. In this case, the glial cells form a mesh-like structure between the skin's outer and inner layers, with filament-like protrusions that extend into the skin's outer layer.
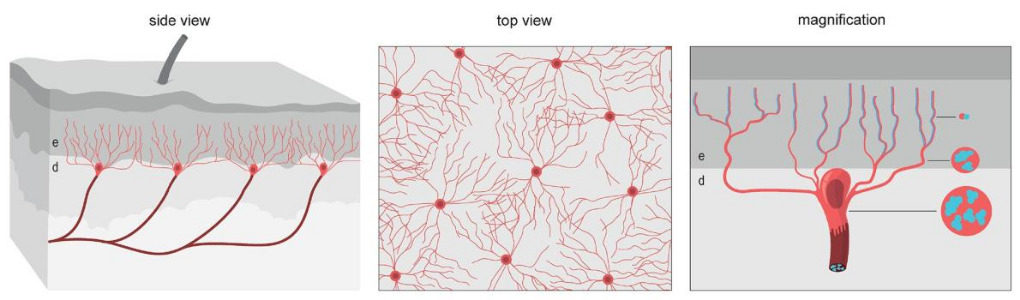
"We have been thinking for probably a hundred years that pain is started from nerves in the skin," says study coauthor Patrik Ernfors, a molecular neurobiologist at the Karolinska Institutet in Sweden. "But what we show now is that pain can also be started in these glial cells."
Pain progression
The team first identified this new organ in mice, and they tested its functionality by measuring the rodents' responses to different types of pain. When the cells in the organ were turned off via gene editing, the animals had a normal response to thermal pain, or discomfort caused by heat or cold. But all of the mice showed a reduced response to mechanical pain when the glial complex was deactivated.
The findings change the way scientists think about how pain begins and progresses — at least in mice. The scientists have not yet checked that the organ exists in humans, but Ernfors says the probability is high.
"Considering that all other previously known sensory organs in [mice] also exist in humans, it is possible if not likely that this sensory organ also is present in our skin," he says.
If so, the work may help inform treatments for a variety of neuropathic pain disorders, which affect an estimated 10 percent of the overall population in the U.S. and between 7 and 10 percent of the population in Europe, Ernfors says. For example, finding ways to alter this cell network may benefit people who suffer from allodynia, a condition in which the skin becomes so sensitive that even a light touch or brush can be excruciating.
"This is a very appealing discovery," says Luana Colloca, a neurophysiologist and an associate professor at the University of Maryland School of Medicine who was not involved with the study. "It's exciting to know that there is a system that is much more than the nociceptive fibers that we teach about to our students."



Just wondering.