What becomes of the broken-hearted? In cardiac medicine, the answer is usually brutally straightforward: they die. Heart disease is the leading cause of death worldwide and there is often precious little we can do about it. Pacemakers bring some relief and transplants work, but there are nowhere near enough donated hearts to go around. And unlike skin and liver cells, heart muscle cells can't remake themselves. Once they get damaged or die, they are gone forever.
Lab-grown stem cells, once the great hope for mending hearts, have disappointed. But over the past few years, cell biologists have been quietly exploring an alternative approach. Rather than growing cells in a dish and transplanting them, they want to switch their identities inside the body, so that we can heal ourselves from within.
That might sound rather fanciful. But cells are proving more malleable than we ever imagined, and now plans are being drawn up for the first human clinical trials to see if we can repair damaged hearts this way. If we can perfect the tricks needed to safely switch cell identity in situ - and it is a big if - we should be able to repair tissues ravaged by all sorts of conditions, from diabetes to dementia.
For a long time after biologists worked out how a featureless cluster of identical cells can become the rich diversity of parts that comprise a body, they generally assumed that adult cells were stuck with their fates. Once embryonic stem cells, capable of becoming any tissue type, had differentiated into skin cells, heart muscle cells, neurons or whatever, there seemed to be no turning back.
But in 2006, Shinya Yamanaka, then at Kyoto University in Japan, showed otherwise in spectacular fashion. He transformed adult mouse cells back into a stem-cell-like state by inserting a cocktail of proteins called transcription factors. These work by changing which of the cells' genes are expressed, switching the cells to a "pluripotent" state in which they are able to differentiate into any tissue in the body. A year later, Yamanaka repeated the trick with human cells, a breakthrough that earned him a share of the Nobel prize for medicine in 2012.
Switching Fates
Yamanaka's induced pluripotent stem (iPS) cells promised a new era of medicine. They provided a means to skip over the ethical quagmire associated with acquiring stem cells from discarded embryos. They also offered an abundant source of therapeutic cells that the immune system shouldn't reject because they could be grown from the transplant patients themselves. The hope was that cells, tissues and perhaps entire organs would soon be grown in the lab from mature cells from a patient, before being transplanted back to repair injury or cure disease.
More than a decade after Yamanaka's discovery, however, iPS cells have yet to deliver. Only a handful of stem-cell therapies have been approved by the US Food and Drug Administration - for several blood disorders, including leukemia, and skin growth after burns - and all of those make use of embryonic stem cells, not iPS cells.
The failure has been felt acutely in the world of cardiology. "No cell-based therapy is close to being approved for heart disease," wrote Roberto Bolli, a cardiologist at the University of Louisville in Kentucky, in response to the latest high-profile failure in February last year. "A rising tide of skepticism has bedeviled the field, leading some critics even to question whether clinical studies should continue."
To some extent that's not surprising. The promise was undoubtedly overhyped to begin with, and it usually takes a long time to get from discovery to clinical use. But there are several reasons why iPS cells haven't made the grade. For one thing, there are still questions about how closely they resemble real stem cells, and so how versatile and safe they are. Then there is the fact that lab-grown cells, once transplanted, struggle to integrate with surrounding tissue.
"You don't need to wipe the slate clean to change the identities of our cells"
Some organs are more amenable to transplantation after in vitro growth because they don't have to integrate, says Deepak Srivastava at the Gladstone Institutes in San Francisco. Insulin-producing pancreatic cells, for instance, can make this hormone anywhere in the body, he says. "But others, such as heart cells, have to integrate closely, so attempts to transplant them after in vitro growth haven't been so successful."
The setbacks spurred Srivastava and others to start exploring an alternative approach that doesn't involve grafting lab-grown cells. The idea is to exploit the plasticity of cells like never before by transforming them inside the body rather than reverting to a pluripotent state first. And you have to do this directly, because iPS cells made inside animals have a tendency to turn cancerous.
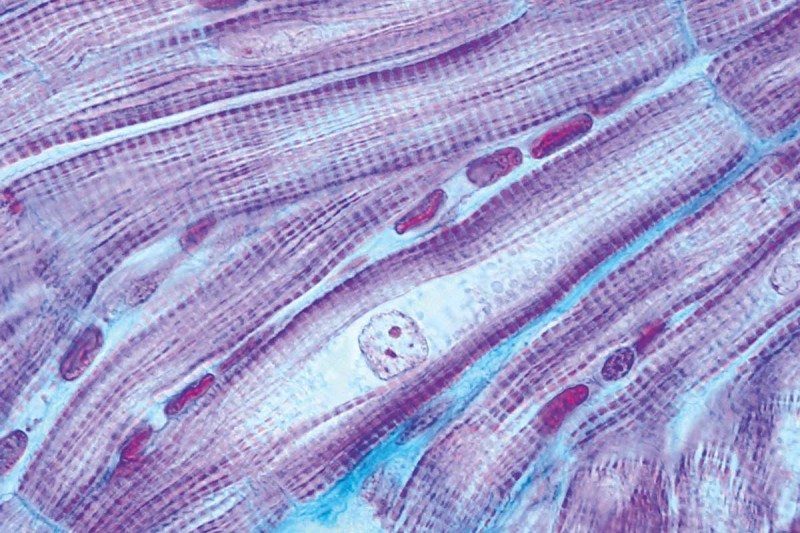
Srivastava was the first to demonstrate such "direction conversion" of heart cells in 2010, when he and his colleagues used just three transcription factors to transform mouse heart fibroblasts, which make up some of the fabric of the organ, to beating heart muscle cells or cardiomyocytes. It was a big surprise, says Srivastava: "Initially there was great scepticism that a small group of factors could switch cell fate." Later, his team went one better by getting those cells to beat in a coordinated fashion, just as in real cardiac muscle. In 2013, they pulled off the trick in human cells too.
Clearly, then, you don't need to wipe the slate clean to transform cells. The trouble is, Srivastava and his colleagues only did all this in cells grown in a Petri dish, where it is easy to deliver the reprogramming agents and control the environment in which they grow. But to avoid the complications associated with transplantation, we need to do it inside the body. This is where things get trickier.
We already have the means to deliver genetic material encoding the proteins needed for transformation, using methods developed for gene therapy. Typically, the genes are delivered by a virus that does not integrate any of its own DNA into target cells' genomes.
Several groups have now used such methods to directly convert cells inside animals. In 2008, for instance, a team led by Douglas Melton at the Harvard Stem Cell Institute transformed ordinary pancreatic cells in mice into insulin-producing beta cells, those destroyed by an autoimmune response in people with type 1 diabetes. Sure enough, the converted cells produced insulin - an encouraging proof of principle.
But making insulin-producing cells is probably one of the easier hurdles to clear for in vivo cell reprogramming. Even if you make them from liver cells, as Jonathan Slack, then at the University of Minnesota, and co-workers have done, the switching required is relatively minor because the two types of cell come from the same precursor cells in the growing embryo. What's more, these beta cells don't have to fully assimilate with the cells around them to produce insulin.
The same cannot be said for heart muscle cells, says Srivastava - they need to coordinate their pulsing activity with the rest of the heart. Nevertheless, in 2012, his team managed to reprogram mouse heart fibroblasts into muscle cells in vivo with three transcription factors. The mice had suffered a heart attack, producing scar tissue that impaired cardiac function. But a couple of months after the treatment, their hearts had begun to recover: some of the scar tissue had been converted into beating heart muscle cells and the organ's ability to pump blood had improved.
Intriguingly, the three proteins that did the trick here don't work on cells in a dish, which suggests that surrounding tissues in the body can make in vivo reprogramming more effective. "There's a general feeling that reprogramming does work better in vivo," says Slack, now at the University of Bath, UK.
It is not entirely clear why, but Sheng Ding at the Gladstone Institutes suspects it could be due to cells that surround the ones being altered supplying chemical or mechanical signals that help the process along. That, he says, is the kind of advantage conferred by the tissue regeneration that occurs naturally in animals such as salamanders, which can famously regenerate entire limbs.
There seems to be no fundamental reason why this reprogramming process can't work in humans, too - and not just for heart disease (see "Regain your brain"). The questions now are how and when that will happen. Ultimately, like any experimental medicine, there are lots of boxes to tick before we can test this new direction in cell reprogramming in humans - and plenty of potential pitfalls.
One obvious concern is that, once injected into the body, gene-tweaking proteins might run amok, drifting away from the place they are supposed to work and transforming other cells elsewhere. In theory, this off-target meddling could happen, says Srivastava, but people don't tend to see it in experiments on animals because "everything has to be just right" to reprogram cells. "The experience of gene therapy indicates that it is very hard to get enough genes into even the target tissue, so off-target effects may not be a big risk," says Slack.
New Growth
"If you have to change cell fates by injecting viruses, that automatically puts you in a more daunting regulatory regime," says Ben Stanger at the University of Pennsylvania in Philadelphia. "What might the virus do?" That is part of the reason why none of the in situ reprogramming breakthroughs over the past few years have led to clinical trials. Melton, for instance, has chosen instead to pursue iPS cells as a treatment for type 1 diabetes.
It is also why people are keen to explore virus-free alternatives, most notably the use of small synthetic molecules instead of genes to do the reprogramming. Srivastava's team, for example, has converted human cardiac cells with small molecules of this kind. The long history of using such compounds as drugs should make it easier to get them through the regulatory system, says Stanger.
On the other hand, the screening process to find the right molecules is expensive, and we don't yet know how to deliver them to the right spot for long enough. But Juan Carlos Izpisua Belmonte of the Salk Institute in La Jolla, California, thinks this approach can bear fruit in the near future. "These chemicals could be administered in creams or injections to rejuvenate skin, muscle or bones," he says.
San Francisco-based Tenaya Therapeutics, spun out of work done at the Gladstone Institutes, is already forging ahead with gene-based reprogramming. According to chief scientific officer Tim Hoey, the company is aiming to switch cells in vivo with genes delivered by viruses, to restore function in hearts that have lost cardiac muscle after a heart attack. Hoey says preclinical studies are under way in animals to determine safety and efficacy ahead of possible human trials.
In the meantime, Srivastava has been pursuing what could be an even better way to repair tissue from the inside: not by repurposing existing cells, but by stimulating the growth of new ones. Earlier this year, he and his colleagues found they could trigger the multiplication of cardiomyocytes in adult mice with yet another mixture of genes, this time ones normally involved in regulating the "cell cycle" that governs proliferation during the gestation of the fetus.
When the team injected the genes into adult mice with damaged hearts, the animals generated fresh heart muscle, something that would never normally happen. Srivastava says it is the first time anyone has been able to get adult cells to start dividing anew with sufficient efficiency to make the approach seem viable for clinical use.
The implications could be profound. If this approach could be made to work for humans too, then ultimately it may not be necessary to repair and replace tissues by putting existing cells to new uses. No changes of identity would be necessary. We could simply persuade our broken hearts, and perhaps even our degenerating brains, to grow again.
Regain Your Brain
Adult brains can't produce enough new brain cells to repair damage caused by stroke or to arrest the progression of neurodegenerative disorders such as Alzheimer's and Parkinson's disease.
That's why the ability to transform adult cells into other cell types inside the body is so appealing. If we can convert other types of brain cell directly into neurons and other nerve cells inside the brain and spinal column, there would be no need to transplant lab-grown cells, which don't tend to integrate with surrounding tissues. It would also bypass the need to drill a hole in the skulls of patients, which is how the researchers behind the most advanced stem-cell therapies for stroke managed to deliver new cells into the brain.
Animal studies are proving promising. In 2013, a team led by Malin Parmar at the University of Lund in Sweden transformed glial cells, which are abundant in the brain, into neurons in rats. A year later, Chun-Li Zhang at the University of Texas Southwestern Medical Center and his colleagues showed that glial cells can be transformed into neuron-forming cells called neuroblasts inside the spinal cords of adult mice.
So far, no one has managed to get new nerve cells to coordinate their activity with the existing network, so they don't help restore nerve function. It is also hard to know whether you are making the right kind of neuron. If such obstacles can be overcome, however, the technique could help us treat spinal column injuries, stroke damage and perhaps even neurodegenerative diseases.
About The Author
Philip Ball is a science writer based in London. His latest book is Beyond Weird (Bodley Head)



If it is heart muscle cells or neurons, this may be electrical signal more responsible than any of these. Though some scientists may consider the electrical signal a chemical one, since gap junction (a.k.a. electrical synapse) involves movement of ions, there are some important functional differences between these, and proper chemical synapses. Heart contains electrical synapses between pacemaker cells and normal cardiomyocytes for example.
Electrical synapses do not offer the fine control of chemical synapses, so connections using these can be overlooked, but they are used by organism when two factors count: speed and coordination of multiple cells (they can transfer signal in both directions, for instance).