
The cloud is located in Titan's stratosphere and is made of a compound known as dicyanoacetylene (C4N2), an ingredient in the chemical cocktail that colors the moon's brownish-orange atmosphere.
Decades ago, NASA's Voyager 1 spacecraft spotted an ice cloud just like this one. What has puzzled scientists ever since is this: they detected less than 1% of the dicyanoacetylene gas needed for the cloud to condense.
Recent observations from NASA's Cassini mission yielded a similar result. Using the Cassini Composite Infrared Spectrometer (CIRS), the scientists found a large high-altitude cloud made of the same frozen chemical.
Yet, just as Voyager found, when it comes to the vapor form of this chemical, CIRS reported that Titan's stratosphere is as dry as a desert.
"The appearance of this ice cloud goes against everything we know about the way clouds form on Titan," said CIRS co-investigator Dr. Carrie Anderson, from NASA's Goddard Space Flight Center.
A different condensation process takes place in the stratosphere — the region above the troposphere — at Titan's north and south winter poles. In this case, layers of clouds condense as the global circulation pattern forces warm gases downward at the pole. The gases then condense as they sink through cooler and cooler layers of the polar stratosphere.
Either way, a cloud forms when the air temperature and pressure are favorable for the vapor to condense into ice. The vapor and the ice reach a balance point that is determined by the air temperature and pressure. Because of this equilibrium, scientists can calculate the amount of vapor where ice is present.
"For clouds that condense, this equilibrium is mandatory, like the law of gravity," said Dr. Robert Samuelson, also from NASA's Goddard Space Flight Center.
But the numbers don't compute for the cloud made from dicyanoacetylene. The scientists determined that they would need at least 100 times more vapor to form an ice cloud where the cloud top was observed by the CIRS instrument.
One explanation suggested early on was that the vapor might be present, but Voyager's instrument wasn't sensitive enough in the critical wavelength range needed to detect it.
But when CIRS also didn't find the vapor, the researchers proposed an altogether different explanation.
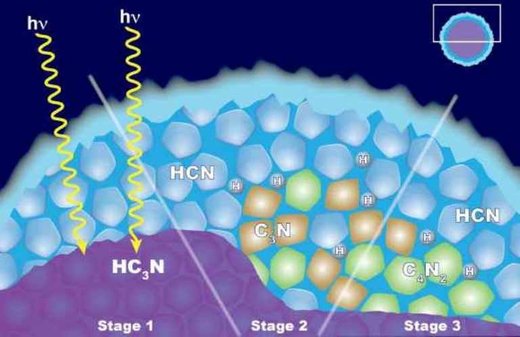
Instead of the cloud forming by condensation, they think the C4N2ice forms because of reactions taking place on other kinds of ice particles. They call this 'solid-state chemistry,' because the reactions involve the ice, or solid, form of the chemical.
The first step in the proposed process is the formation of ice particles made from the related chemical cyanoacetylene (HC3N).
As these tiny bits of ice move downward through Titan's stratosphere, they get coated by hydrogen cyanide (HCN). At this stage, the ice particle has a core and a shell comprised of two different chemicals. Occasionally, a photon of UV light tunnels into the frozen shell and triggers a series of chemical reactions in the ice. These reactions could begin either in the core or within the shell. Both pathways can yield dicyanoacteylene ice and hydrogen as products.
The researchers got the idea of solid-state chemistry from the formation of clouds involved in ozone depletion high above Earth's poles.
"It's very exciting to think that we may have found examples of similar solid-state chemical processes on both Titan and Earth," Dr. Anderson said.
The team suggests that, on Titan, the reactions occur inside the ice particles, sequestered from the atmosphere. In that case, dicyanoacetylene ice wouldn't make direct contact with the atmosphere, which would explain why the ice and the vapor forms are not in the expected equilibrium.
The team's findings were published in the April 16 issue of the journal Geophysical Research Letters.
C.M. Anderson et al. 2016. Solid-state photochemistry as a formation mechanism for Titan's stratospheric C4N2 ice clouds. Geophysical Research Letters 43 (7): 3088-3094; doi: 10.1002/2016GL067795
This article is based on a press-release issued by NASA.



more believable if the scientists had first explained how crop circles are created on Earth. Oak Ridge scientists had investigated and now know a transmutation takes place which leaves radioactivity behind.
Now that we are leaving 'chemicals' behind and progressing fitfully into biologics--India and China are established producers ---we should be very cautious of claiming yet another 'chemical' reaction that may prove outdated before it is believed.