V-safe is a smartphone-based tool that uses text messaging and web surveys to provide personalised health check-ins after receiving a covid injection. The after-vaccination health checker app was developed and implemented to monitor covid-19 "vaccine" safety and as an active surveillance supplement to existing CDC vaccine safety monitoring programs. It was launched on 13 December 2020, the day before covid injections were first made available to the American public.
V-safe has been used by the CDC to monitor covid-19, monkeypox or mpox, and now respiratory syncytial virus ("RSV") vaccines.
Last year, after months of litigation, a US court ordered the CDC to release the "check-the-box" data. You can find the data released so far HERE.
But as Siri discovered, the "check-the-box" data omitted vital information concerning the safety of the covid injections.
Some V-safe questions that users are asked to answer had "check-the-box" options and a "free-text" field to enter additional information. The number of characters in these fields is limited to 250. To indicate what 250 characters look like, this paragraph contains 282 characters.

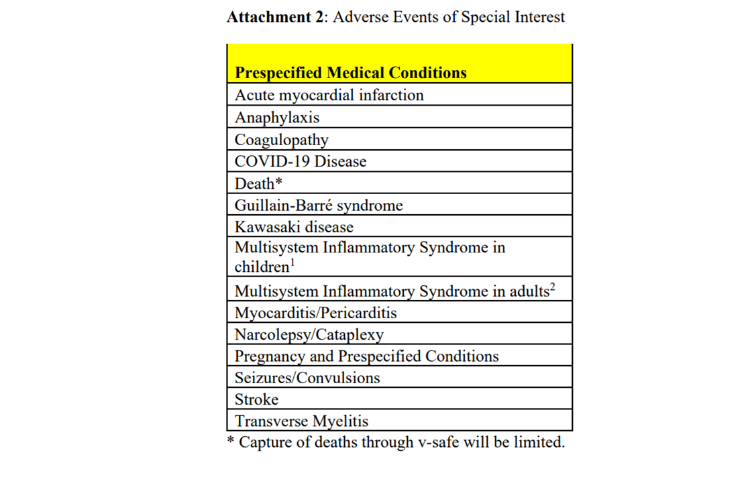
Below is a screenshot taken from page 50 of the CDC's V-safe protocols showing the adverse events of special interest identified but which are not included in "check-the-box" options.
"The adverse events of special interest listed in [the chart above] included myocarditis, pericarditis, acute myocardial infarction, stroke, GBS, and transverse myelitis, among other events," Siri wrote.
"V-safe was launched without, and was never updated, to include any 'check-the-box' fields for these conditions ... It instead relegated them to only potentially be captured in 'free-text' fields!"
By not including the adverse events of special interest as "check-the-box" options and forcing users to write them in the "free-text" fields, the CDC hid these adverse events and so avoided being able to easily calculate a rate of harm.
So, on behalf of the Freedom Coalition of Doctors for Choice, Siri, who also regularly represents the Informed Consent Action Network ("ICAN"), took the matter to court to request that the information from the "free-text" fields be released.
On 5 January, the judge ordered that the CDC release the "free-text" information over the next 12 months. Announcing the news, Siri said: "In our prior case, we obtained the V-safe 'check-the-box' data and, now through this case, the V-safe 'free-text' data will also be made available to the public."
Siri extracted some highlights from the judge's "incredible decision," some of which we have reproduced below:
Rapid vaccination of a huge percentage of the American population is nothing short of astounding, and the endeavour continues ... As of 11 May 2023, the CDC reports that more than 81% of Americans have received at least one dose, including nearly 32 million children. Understandably, there is substantial public interest in the data that supported, and continues to support, the government's promotion of the covid-19 vaccines and boosters.As ordered by the judge, the information will be released in 12 tranches. The first batch of data is due to be released by 15 February 2024. The final tranche is due to be released by 15 January 2025.
Plaintiff [the Freedom Coalition of Doctors for Choice] has shown an urgent need to inform the public about "actual or alleged Federal Government activity" - namely, related to the health and safety of the covid-19 vaccines and policies....
Additionally, as addressed above, Plaintiff presents evidence that calls into question the claim that the vaccines are safe and effective - or at least the scope of research supporting that claim ...
Notably, the sample size is massive - representing between 3-4.5% of the vaccinated population - thus permitting particularly accurate research. The V-safe free-text responses will contribute to the public's understanding of the covid-19 vaccines - specifically as to the assertion by Defendants [the CDC], the Biden administration, and others that the vaccine is "safe and effective" for everyone over six months of age - by providing access to the direct source material ...
Further, all Americans continue to be the target audience of marketing and messaging campaigns to promote continued vaccination.
V-Safe Part 10: Federal Judge Orders CDC to Make Public 7.8 Million V-safe Free-Text Entries Within 12 Months, Aaron Siri, 11 January 2024 as extracted from the Memorandum Order And Opinion issued by Judge Matthew J. Kacsmaryk for the District Court For The Northern District Of Texas Amarillo Division




Why is the information being dripped out in monthly installments?