The first, published in the December 2018 issue of Frontiers in Psychiatry,1 focused on the microbiome's role in insomnia and depression. As noted by the authors:
Numerous studies have suggested that the incidence of insomnia and depressive disorder are linked to biological rhythms, immune function, and nutrient metabolism, but the exact mechanism is not yet clear.Your Gut Microbiome's Role in Insomnia and Depression
There is considerable evidence showing that the gut microbiome not only affects the digestive, metabolic, and immune functions of the host but also regulates host sleep and mental states through the microbiome-gut-brain axis.
Preliminary evidence indicates that microorganisms and circadian genes can interact with each other. The characteristics of the gastrointestinal microbiome and metabolism are related to the host's sleep and circadian rhythm.
Previous studies have shown your gut microbiome can play a significant role in depression and anxiety, and that your diet — which can effectively alter the bacterial composition of your gut — can raise or lower your risk of these mood disorders.
As noted in the Frontiers in Psychiatry study,2 mounting research suggests your gut microbiome helps regulate not only your mood but also your sleep cycle through what's known as the gut-brain axis — a bidirectional communication "highway" that links your central and enteric nervous systems.3
According to the authors of this paper, your gut microbiota affect brain function through three different pathways, all of which have this bidirectional flow:4
- The immunoregulatory pathway — Here, gut bacteria influence brain function via interaction with immune cells that regulate your levels of cytokines, cytokinetic reaction factor and prostaglandin E2.
- The neuroendocrine pathway — With more than 20 types of enteroendocrine cells, your intestine is the largest endocrine organ in your body. As explained by the authors,5 "The gut microbiome may affect the hypothalamic-pituitary-adrenal (HPA) axis and the central nervous system (CNS) by regulating the secretion of neurotransmitters such as cortisol, tryptophan and serotonin (5-HT)."
- The vagus nerve pathway — Your enteric nervous system also plays an important role in the vagus nerve pathway. Here, gut microbiota can exert influence via sensory neurons in your intestinal myenteric plexus.
The authors note that neurotoxic metabolites produced by various gut microbiota can also enter into your CNS via your vagus nerve, "thereby affecting brain function, stress responses and sleep structure."
As mentioned, the information flow through these three pathways is bidirectional, so your CNS can also regulate the composition of your gut microbiome through these pathways.
As one example, through its ability to alter the function of epithelial cells in your intestine, the HPA axis can affect the bacteria's living environment, and in so doing, influence the composition of your gut microbiome.6
Circadian Genes Affect Your Gut Microbiome
In 2017, the Nobel Prize in Physiology or Medicine went to three biologists for their discovery of master genes that control your body's circadian rhythms. Your body contains not just one biological clock, but a vast array of clocks that regulate everything from metabolism to psychological functioning.
While the master clock in your brain synchronizes your bodily functions to match the 24-hour light and dark cycle, each and every organ, indeed each cell, has its own biological clock.
Half of your genes are also under circadian control, turning on and off in cyclical waves. As you might expect, the microbiota in your gut are also under circadian control, which means disruptions in sleep can affect the composition and health of your microbiome as well. As reported in Frontiers in Psychiatry:7
Evidence suggests that Clostridiales, Lactobacillales, and Bacteroidales, which account for ~60% of the microbiota, show significant diurnal fluctuations that result in time-of-day-specific taxonomic configurations.So, to summarize the findings of this Frontiers in Psychiatry, there's a bidirectional connection between your gut microbiome, your sleep and your risk of depression, and endocrine hormones and clock genes play important roles in these processes.
Liang et al. found that the two primary components of the mammalian intestinal microbiota, Bacteroidetes, and Firmicutes, showed cyclical changes in abundance from day to night that are related not only to rhythmic food intake and dietary structure but also to the biological clock and gender of the host.
Recent studies showed that circadian clock misalignment, sleep deprivation, and shift experience changes circadian clock gene expression and microbial community structure.
Interfering with the sleep patterns of mice can also change the structure and diversity of the intestinal microbiota. These findings suggest that circadian genes might affect the intestinal microbiota.
Microbiome Diversity Linked to Sleep Quality
The second study8,9 addressing the curious link between your gut health and sleep was published in PLOS ONE in 2019. Here, the researchers investigated how the microbes in your gut affect the actual quality of your sleep — which we already know can have far-reaching effects on your general health.
Poor sleep and/or lack of sleep has been linked to a wide variety of ailments and health conditions, ranging from poor cognitive performance and neurological dysfunction to an increased risk for Type 2 diabetes,10 heart disease,11 cancer12 and Alzheimer's disease.13
On the other hand, high-quality sleep is associated with improved health, cognition and increased creativity, as discussed in "Using Sleep as a Tool for Creativity."
Using advanced sleep measuring devices, the researchers were able to measure the quality of participants' sleep, which was then compared to the composition of their gut microbiome to see if a correlation could be made. As reported by the authors:14
We found that total microbiome diversity was positively correlated with increased sleep efficiency and total sleep time, and was negatively correlated with wake after sleep onset. We found positive correlations between total microbiome diversity and interleukin-6, a cytokine previously noted for its effects on sleep.The Role of Cytokines
Analysis of microbiome composition revealed that within phyla richness of Bacteroidetes and Firmicutes were positively correlated with sleep efficiency, interleukin-6 concentrations and abstract thinking.
Finally, we found that several taxa (Lachnospiraceae, Corynebacterium, and Blautia) were negatively correlated with sleep measures. Our findings initiate linkages between gut microbiome composition, sleep physiology, the immune system and cognition. They may lead to mechanisms to improve sleep through the manipulation of the gut microbiome.
As mentioned at the beginning, sleep is known to influence your immune function, and cytokines, produced in response to an antigen, are multifunctional chemical messengers that help regulate your innate and adaptive immune systems.15
Interestingly, cytokines also appear to act as a "critical interface between sleep physiology and gut microbiome composition," according to this study. As explained by the authors:16
The acute phase pathway cytokines IL-1β and IL-6 in particular are strongly associated with sleep physiology. IL-1β is a major somnogenic factor. IL-1β administration in human and non-human animals increases spontaneous sleep and fatigue, and IL-1β increases with ongoing sleep loss.So, in summary, what this PLOS ONE study reveals is that the composition of your gut microbiome, the quality and quantity of your sleep, your immune function and cognition are all connected.
Unlike IL-1β, IL-6 is not a direct somnogenic factor, but sleep loss results in increased IL-6 levels. In the gut, IL-6 and IL-1β mediated-inflammation fluctuate in response to stress and disease.
For example, intestinal mucositis results in increased expression of IL-6 and-IL-1β in the small intestine and in serum and colon tissue in mice. In humans, chronic stress alone increases IL-6 and-IL-1β.
Despite the close relationship between cytokine activity, gut microbiome activity and sleep, only a handful of studies have examined sleep and gut-microbiome composition ... In humans, previous research has shown that partial sleep deprivation can alter the gut microbiome composition in as little as 48 hours ...
A more recent study showed that high sleep quality was associated with a gut microbiome containing a high proportion of bacteria from the Verrucomicrobia and Lentisphaerae phyla, and that this was associated with improved performance on cognitive tasks.
Lack of Sleep Makes Chronic Health Problems Extra Risky
The finding that poor sleep and lack of sleep can deteriorate your gut health helps explain why sleeping too little when you're struggling with a chronic health issue could be a downright deadly prescription. As reported by CNN Health:17
If you're a middle-aged adult with high blood pressure, Type 2 diabetes or existing heart disease and you typically sleep less than six hours each night, you could be setting yourself up for cancer or an early death from heart disease.The study18,19,20 CNN is referring to was published in the October 2019 issue of the Journal of the American Heart Association (JAHA). In it, researchers sought to determine whether short sleep duration would increase the risk of death associated with cardiometabolic risk factors and cardiovascular and cerebrovascular diseases.
Data from 1,654 adults from the Penn State Adult Cohort were evaluated. Using Cox proportional hazard models, the adjusted hazard ratio for all-cause mortality among those who slept less than six hours and had cardiometabolic risk factors (high blood pressure, elevated glucose or Type 2 diabetes) was 2.14 times higher than those who regularly slept six hours or more.
They also had a 1.83 times higher risk of dying from cardiovascular or cerebrovascular diseases. Among those with a diagnosis of heart disease or stroke, sleeping less than six hours a night increased their all-cause mortality risk by 3.17 times. Interestingly, it also increased their risk of dying from cancer, specifically, by 2.92 times.
All of these associations were found to be independent of age, sex, ethnicity, obesity, smoking and other health conditions that might influence the results. Conversely, sleeping less than six hours did not increase the risk of death in those that did not have cardiometabolic risk factors or a cardiovascular or cerebrovascular disease diagnosis.
Likewise, those with cardiometabolic risk factors or a cardiovascular or cerebrovascular disease diagnosis who slept six hours or more were not at increased risk for death either. It was specifically the combination of chronic health problems and short sleep duration that increased the risk of death, including cancer mortality.
Sleep Duration Plays a Role in Mortality Prognosis
As noted by the authors:21
Our novel findings show that objective short sleep duration increases the mortality risk of middle‐aged adults with CMRs [cardiometabolic risk factors] and those who have already developed CBVD [cardiovascular and cerebrovascular diseases].General Sleep Guidelines
Middle‐aged adults with CMR who slept <6 hours were at a high risk of dying from CBVD, whereas middle‐aged adults with CBVD who slept <6 hours were at a high risk of dying from cancer ...
If these findings are replicated in other large cohorts with objective sleep measures, short sleep duration should be included in the prediction of the mortality prognosis of middle‐aged adults with CMR or CBVD.
The primary finding of the current study indicated that there was an ≈2‐fold risk for all‐cause, CBVD, and non‐CBVD mortality in participants who had CMRs at baseline and demonstrated short sleep duration in the sleep laboratory.
Individuals who had CMRs and normal sleep duration at baseline, on the other hand, did not show a significantly increased risk on any of the mortality outcomes. This finding suggests that obtaining an adequate amount of sleep may minimize the adverse effect of CMRs on multiple mortality outcomes.
For instance, participants with both CMRs and short sleep at baseline showed an 83% higher risk of dying from CBVD, whereas their CMR counterparts with normal sleep duration had a modest 35% nonsignificant higher risk of CBVD mortality ...
In conclusion, objective short sleep duration is an effect modifier of the mortality risk associated with CMR or CBVD. More important, our data suggest that short sleep may operate through different mechanisms on CBVD versus cancer mortality.
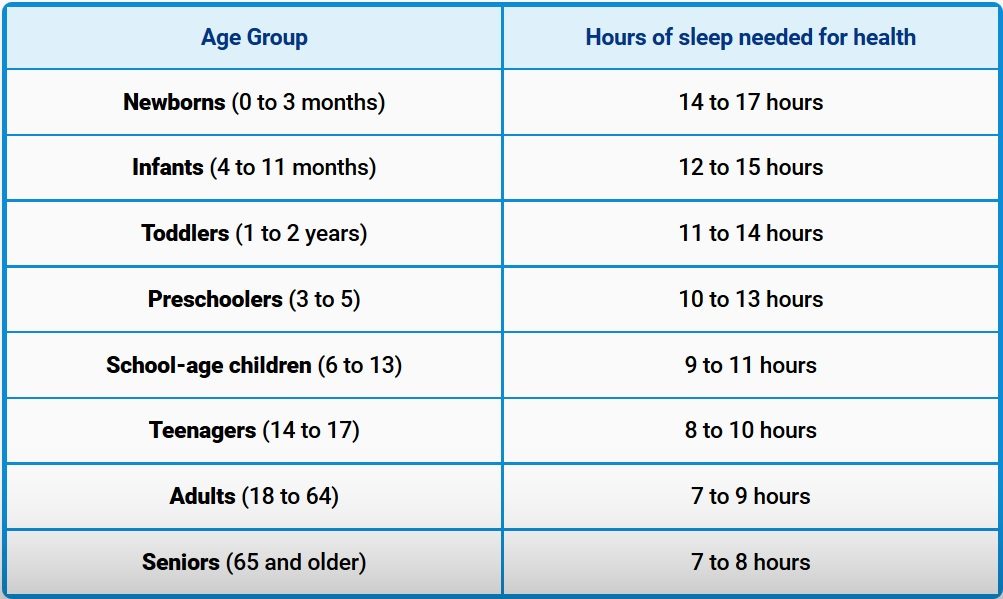
Considering the massive importance of sleep for preventing the two top killers in the U.S. (heart disease and cancer), just how much sleep do you need to reap protective benefits?
According to a scientific review of more than 300 studies published between 2004 and 2014, a panel of experts came up with the following recommendations. Keep in mind that if you're sick, injured or pregnant, you may need a bit more than normal.
Set a Nightly Alarm to Help You Get Enough Sleep
There's simply no doubt that sleep needs to be a priority in your life if you intend to live a long and healthy life. For many, this means forgoing night-owl tendencies and getting to bed at a reasonable time.
If you need to be up at 6 a.m., you need a lights-out deadline of 9:30 or 10 p.m., depending on how quickly you tend to fall asleep. If you find it difficult to get to bed on time, consider setting a bedtime alarm to remind you that it's time to shut everything down and get ready for sleep. For further guidance, see "Sleep — Why You Need It and 50 Ways to Improve It."
How to Nurture Your Gut Microbiome
As for how to improve and maintain a healthy microbiome — which might do more to improve your sleep than is currently appreciated — it isn't very complicated, but you do need to take proactive steps to encourage its health while avoiding factors known to cause harm. This includes:
Do: Eat plenty of fermented foods — Healthy choices include lassi, fermented grass fed kefir, natto (fermented soy) and fermented vegetables.Sources and References
Avoid: Antibiotics, unless absolutely necessary, and when you do, make sure to reseed your gut with fermented foods and/or a high-quality probiotic supplement.
Do: Take a probiotic supplement — Although I'm not a major proponent of taking many supplements (as I believe the majority of your nutrients need to come from food), probiotics are an exception if you don't eat fermented foods on a regular basis.
Avoid: Conventionally raised meats and other animal products, as factory farmed animals are routinely fed low-dose antibiotics and GE grains loaded with glyphosate, which is widely known to kill many bacteria.
Do: Boost your soluble and insoluble fiber intake, focusing on vegetables, nuts and seeds, including sprouted seeds.
Avoid: Chlorinated and/or fluoridated water — Especially in your bathing such as showers, which are worse than drinking it.
Do: Get your hands dirty in the garden — Exposure to bacteria and viruses can help to strengthen your immune system and provide long-lasting immunity against disease.
Getting your hands dirty in the garden can help reacquaint your immune system with beneficial microorganisms on the plants and in the soil.
Avoid: Processed foods — Excessive sugar feeds pathogenic bacteria.
Food emulsifiers such as polysorbate 80, lecithin, carrageenan, polyglycerols and xanthan gum also appear to have an adverse effect on your gut flora.
Unless 100% organic, they may also contain GMOs that tend to be heavily contaminated with pesticides such as glyphosate. Artificial sweeteners have also been found to alter gut bacteria in adverse ways.22
Do: Open your windows — For the vast majority of human history, the outside was always part of the inside, and at no moment during our day were we ever really separated from nature.
Today, we spend most of our lives indoors. And, although keeping the outside out does have its advantages, it has also changed the microbiome of your home.
Research shows that opening a window and increasing natural airflow can improve the diversity and health of the microbes in your home, which in turn benefit you.23
Avoid: Agricultural chemicals — Glyphosate (Roundup) in particular is a known antibiotic and will actively kill many of your beneficial gut microbes if you eat foods contaminated with it.
Do: Wash your dishes by hand instead of in the dishwasher — Research has shown that washing your dishes by hand leaves more bacteria on the dishes than dishwashers do, and eating off these less-than-sterile dishes may actually decrease your risk of allergies by stimulating your immune system.
Avoid: Antibacterial soap — It kills both good and bad bacteria and contributes to the development of antibiotic resistance.
- 1, 2 Frontiers in Psychiatry 2018; 9: 669
- 3 Annals of Gastroenterology 2015 Apr-Jun; 28(2): 203-209
- 4, 5, 6 Frontiers in Psychiatry 2018; 9: 669, The Gut Microbiome and the Gut-Brain Axis
- 7 Frontiers in Psychiatry 2018; 9: 669, The Gut Microbiota, Clock Genes and Sleep + Gut Microbiota and Comorbidity of Insomnia and Depression
- 8 PLOS ONE 2019; 14(10): e0222394
- 9 Science Daily October 28, 2019
- 10 JAMA 2005;165(8):863-867
- 11 Archives of Internal Medicine 2003;163(2):205-209
- 12 Sleep Medicine Reviews August 2009; 13(4): 257-264
- 13, 16 PLOS ONE 2019; 14(10): e0222394, Introduction
- 14 PLOS ONE 2019; 14(10): e0222394, Abstract
- 15 Biology LibreTexts, Cytokines Important in Innate Immunity
- 17 CNN Health October 2, 2019
- 18 JAHA October 2, 2019;8:e013043
- 19 Medical News Today October 3, 2019
- 20 Science Daily October 2, 2019
- 21 JAHA October 2, 2019;8:e013043, Discussion
- 22 Scientific American March 17, 2015
- 23 ISME Journal 2012 Aug;6(8):1469-79




Comment: See also: