© Nature NewsResearchers created ultra-light and ultra-heavy forms of hydrogen to probe the laws of quantum chemistry.
Scientists have created ultra-light and ultra-heavy forms of the element hydrogen, and have investigated their chemical properties.
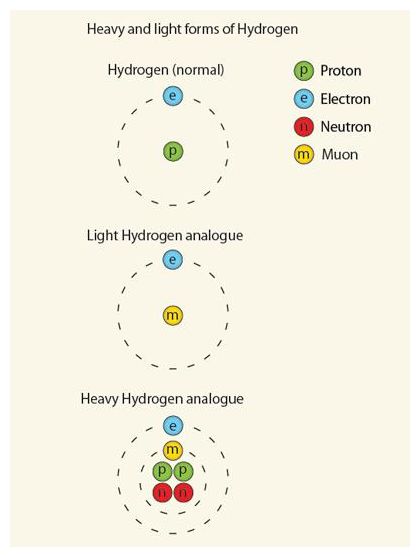
Donald Fleming, a chemist at the University of British Columbia in Vancouver, Canada, and his colleagues generated two artificial analogues of hydrogen: one with a mass a little over one-tenth that of ordinary hydrogen, and one four times heavier than hydrogen. These pseudo-hydrogens both contain short-lived subatomic particles called muons - super-heavy versions of the electron.
The researchers tested the behaviour of these new atoms in a chemical reaction called a hydrogen exchange, in which a lone hydrogen atom plucks another from a two-atom hydrogen molecule - just about the simplest chemical reaction conceivable. In a paper in
Science,1 they report that both the weedy and the bloated hydrogen atoms behave just as quantum theory predicts they should - which is itself surprising.
The experiment is a "tour de force", says Paul Percival, a muonium chemist at Simon Fraser University in Burnaby, Canada.
"I would never attempt such a difficult task myself," he says, "and when I first saw the proposal I was very doubtful that anything of value could be gained from the Herculean effort. Don Fleming proved me wrong. I doubt if anyone else could have achieved these results."




