A new super-resolution microscopy technique is providing researchers with never-before-seen detail of a cell membrane.
They claim the development could open doors to new diagnostic, prevention and treatment techniques.
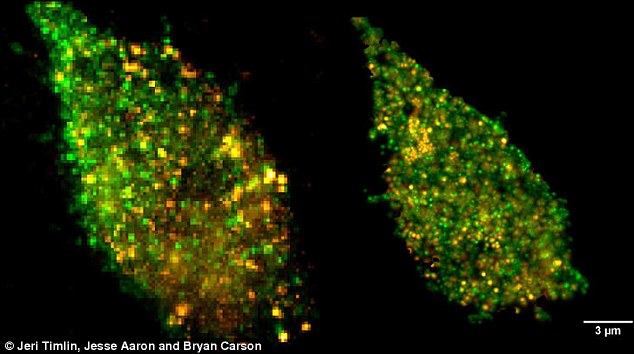
The cell membrane is a bustling hub of activity on a minuscule scale. While providing structure and housing the cell's interior, the membrane regulates movement of materials in and out of the cell, controls adhesion to other objects and co-ordinates the cell's communications and subsequent actions through signalling.
Receptor proteins on the surface of immune cells, known as toll-like receptors (TLRs), are tasked with recognising intruders, or antigens.
The TLR4 member of this receptor family responds to certain types of bacteria by detecting lipopolysaccharides (LPS) present on their surface.
TLR4 proteins then alert the cell and activate an immune response.
Using imaging techniques they developed, Sandia researchers Mr Aaron, Jeri Timlin and Bryan Carson discovered that TLR4 proteins cluster in the membrane when confronted with LPS derived from E.coli, which increases cell signalling and response.
Interestingly, LPS derived from the bacteria that cause plague - Yersinia pestis - do not cause the same effects.
This could explain why some pathogens are able to thwart the human immune system.
The plague studies marked the first time such small events have been imaged and compared, the researchers said.
Previously, even the most sophisticated optical microscopes could not image the cell surface with enough spatial resolution to see the earliest binding events, due to the diffraction barrier, which limits what can be resolved using visible light.
'With more traditional visualisation methods, you can't see the level of detail you need. It's important to look at not only what's present, but also when and where it's present in the cell,' Mr Timlin said.
The technique builds on super-resolution capabilities developed in recent years, but goes another step by adding dual-colour capabilities to the relatively new stochastic optical reconstruction microscopy, or STORM.
The combination enables the Sandia team to get a more complete picture by simultaneously imaging LPS and TLR4 receptors on the membrane.
Mr Carson said: 'Current light microscopy capabilities are akin to looking out the window of an airplane and seeing the irrigation circles. You know that plants are there, but you can't tell what kinds of plants they are or what shape the leaves are.
'But with this technology, it's like zooming in and seeing the leaves and the structure of the plants. That buys you a lot in terms of understanding what's happening within a cell and specifically how the proteins involved interact.'
Next on the team's agenda is developing the capability to image live cells in real-time using spectral Stimulated Emission Depletion.
'We're working toward using a version of super-resolution that's much more live-cell friendly, and extending that in terms of what colours are available to do multiple colours, while maintaining the live-cell friendliness,' Mr Timlin said.
'I see this as a beginning of a long development in this type of imaging technology.'
The technology has exciting potential in immunology and drug discovery. Improved imaging could show the mechanisms viruses use to invade cells, which might lead to drugs that would block entry.
'We're hoping to do something like label the viral particles and watch them in real-time, or as close as we can to real-time, in the internalisation process,' Mr Carson said.
'With the super-resolution technique, we can actually watch them move through the membrane and see if there are other structures being recruited by the virus to the site of internalisation.'



Reader Comments
to our Newsletter