Earth's spin isn't flawless. Geophysicists have discovered that the time it takes our planet to complete one rotation - the length of a day - fluctuates slightly over the course of months or years. They've also noticed extra swing in the predictable wobble of Earth's axis of rotation, like the swaying of a spinning top. The variations are probably caused by the solid iron inner core, liquid metal outer core, and rocky mantle rotating at slightly different rates. Friction helps bring them into line, and the magnetic field of the outer core can pull on the metal inner core. But to really fit the observations, the core should also exert its magnetic tug on the mantle, says Bruce Buffett, an earth scientist at the University of California, Berkeley, who was not involved in the new study. This means that a layer of the mantle must be able to conduct electricity. But, he says, "the origin of the metallic layer remains an open question."
The main components of the mantle rock, iron monoxide included, don't conduct electricity at temperatures and pressures we're used to here at the surface. But research in the 1980s suggested things might be different deep down: An electrical current passed through the material more easily when it was exposed to a shock wave. The pressure of the shock wave compressed the arrangement of iron and oxygen in the iron monoxide, allowing the electrons to travel more freely from atom to atom.
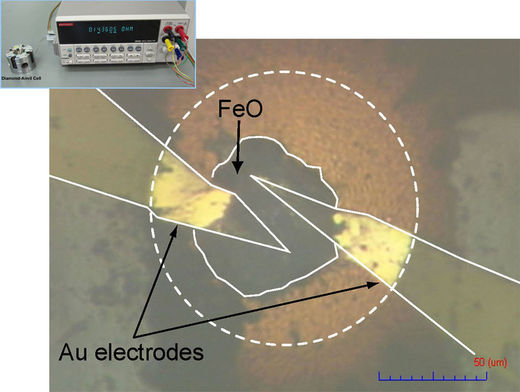
This work piqued the curiosity of Kenji Ohta, who studies materials under extreme conditions at Osaka University in Japan. To find out whether pressure could turn iron monoxide into a conductor in Earth's mantle, Ohta and colleagues heated a disk of the material with a laser and compressed it in a diamond anvil. At the same time, they measured how well it conducted electricity by passing a current through it while also keeping tabs on the arrangement of its atoms with x-rays. At about 700,000 times the pressure of Earth's atmosphere and a temperature of 1600°C, the team found that the iron monoxide conducted electricity as well as a metal.
The iron monoxide had made the transition from nonconductive to conductive, but its structure hadn't changed. Instead, it had made a new kind of transition, says geophysicist Ronald Cohen of the Carnegie Institution for Science in Washington, D.C., who led the computer simulation of the iron monoxide. The change hinges on the material's magnetic properties at high temperatures. In nonconductive iron monoxide, each potentially mobile electron is trapped in an iron atom. It can't move easily through the crystal, but it can align with magnetic fields like a compass needle - that is, it is in a magnetic state. At high temperatures and pressures, the electrons begin to fluctuate between the magnetic state and a nonmagnetic state, in which they no longer respond to a magnetic field. This cuts their ties to their atoms, and they move freely as in a metal, the team reported 12 January in Physical Review Letters.
When the researchers raised the pressure to 1.4 million atmospheres and the temperature to 2200°C, conditions comparable to the interior of Earth, the iron monoxide was still in a metallic form. They then predicted the conductivity at 3430°C - the temperature at the boundary between the mantle and core - and found that iron monoxide remained a respectable conductor.
Buffett points out that the mantle's 9% iron monoxide isn't enough to guarantee that a connected layer will form. It would have to be concentrated near the outer core, possibly making up about 90% of the material there. Buffett notes that other researchers have proposed chemical reactions between the mantle and core that might produce compounds like iron monoxide. "Impressive recent advances in both experiments and theory" could soon evaluate the plausibility of a metallic iron monoxide layer, he says.



Reader Comments
to our Newsletter