
For the oil and gas industry, the substances are also known to be the primary hazard when drilling for deepwater oil.
Methane hydrates are volatile compounds - natural gas compressed into molecular cages of ice. They are stable in the extreme cold and crushing weight of deepwater, but are extremely dangerous when they build up inside the drill column of a well. If destabilized by heat or a decrease in pressure, methane hydrates can quickly expand to 164 times their volume.
Survivors of the BP rig explosion told interviewers that right before the April 20 blast, workers had decreased the pressure in the drill column and applied heat to set the cement seal around the wellhead. Then a quickly expanding bubble of methane gas shot up the drill column before exploding on the platform on the ocean's surface.
Even a solid steel pipe has little chance against a 164-fold expansion of volume - something that would render a man six feet six inches tall suddenly the height of the Eiffel Tower.
Scientists are well aware of the awesome power of these strange hydrocarbons. A sudden large scale release of methane hydrates is believed to have caused a mass extinction 55 million years ago. Among planners concerned with mega-disasters, their sudden escape is considered to be a threat comparable to an asteroid strike or nuclear war. The Lawrence Livermore National Laboratory, a Livermore, California-based weapons design center, reports that when released on a large scale, methane hydrates can even cause tsunamis.
So it is not surprising to anyone who knows about the physics of these compounds that the Deepwater Horizon rig was lost like a waterfly crumpled by a force of nature scientists are still just getting to know.
Number One Deepwater Drilling Issue
SolveClimate contacted scientists at the Colorado School of Mines, Center for Hydrate Research, who focus on the fundamental science and engineering of methane hydrates to gain further insight. They did not want to speculate on the role that methane hydrates could have played in the BP disaster, but they were willing to provide a basic understanding of the nature and behavior of these familiar but little understood substances.
"Gas hydrates are the number one flow assurance issue in deepwater drilling," Carolyn Koh, an associate professor and co-director of the Hydrate Center, told us in an exclusive interview.She explained that the oil and gas industry has a lot of experience with methane hydrates, because they have to be kept from forming in pipes or they will clog the lines, stop the flow of oil, and pose a danger. Drillers use inhibitors such as methanol to keep the hydrates from crystallizing inside drill rigs operating at great depth, where conditions for methane hydrate formation are ideal.
This film clip of an experiment conducted on the ocean floor near the Deepwater Horizon drilling site demonstrates how quickly and easily methane hydrates can form. It was conducted by the Gulf of Mexico Hydrates Research Consortium aboard the Seward Johnson in September 2006. The voices of the scientists conducting the experiment are clearly audible.
The clip shows with remarkable clarity a robotic arm maneuvering a clear tube over a stream of hydrate bubbles emanating from a crater on the sea floor. Within minutes, gas trapped in the tube begins to form a visible solid - a white ice matrix - thanks to the extreme cold and pressure of the ocean depth. When the tube is inverted, the hydrate, less dense than seawater, floats out of the tube, dissociating into its components, gas and water.
Oil and gas drillers encounter far greater volumes of methane hydrate than the gentle stream of bubbles escaping from a small fissure that are shown in the film.
Amadeu Sum, an assistant professor at the Colorado School of Mines and also a co-director of the Hydrate Center, explained that methane hydrates can be encountered by drillers in the deep ocean where methane hydrates are trapped in sediments beneath the ocean floor.
Vast Deposits in Ocean Sediments
Professor Sum explained gas and oil flow up the pipe together in normal drilling operations. These hydrocarbons occur naturally together in conventional drilling operations. The deepwater of the Gulf of Mexico, and other places where methane hydrates exist, present drillers with special safety challenges.
For one thing, methane hydrates are believed to exist in vast deposits underneath the ocean floor, trapped by nature in ocean sediments. Deepwater drillers could find themselves drilling through these natural hydrate deposits.
Professor Sum said geologists know much less about these hydrate-bearing sediments than conventional ocean sediments, and that there is "little knowledge of the risks" of drilling into them.
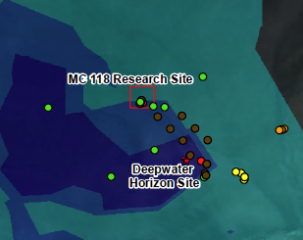
The Deepwater Horizon rig was drilling in Block 252 of an area known as the Mississippi Canyon of the Gulf, thought to contain methane hydrate-bearing sediments, according to government maps. The platform was operating less than 20 miles from a methane hydrate research site located in the same canyon at Block 118.
From the sea floor a mile down, the Deepwater Horizon rig had penetrated another 18,000 feet - almost another five miles down - into the earth's crust with pipe.
According to the National Academy of Sciences, which published a bullish report on the energy potential of methane hydrates,
"Industry practice is to avoid methane-bearing areas during drilling for conventional oil and gas resources for safety reasons."Professor Sum explained that because "with oil there is usually gas present," it is possible for methane hydrates to form in the pipe even when not drilling through hydrate-bearing sediments. The pressure and cold of the deepwater create conditions that encourage gas flowing into the pipe to form hydrates, and if the rate of crystallization is rapid enough, the hydrates can clog the pipe.
The cofferdam that BP lowered over the broken pipe gushing oil to contain the spill was almost immediately clogged by methane hydrates, which formed spontaneously. Gas escaping with the oil from the well, when trapped in the steel structure with cold water under great pressure, rapidly accumulated into an ice-like matrix.
Documented Explosive Hazard
In a book about methane hydrates, which Professor Koh co-authored, brief mention is made of a case in which methane hydrates caused a gas pipe to rupture on land, leading to loss of life.
Two workers were attempting to clear a line in which a hydrate plug had formed. The authors say that "the impact of a moving hydrate mass" caused the pipe to fail. The explosion caused a large piece of pipe to strike the foreman, killing him. The book then quotes from the Canadian Association of Petroleum Producers Hydrate Guidelines to describe proper procedures for safely removing a hydrate plug in a pipe on land.
SolveClimate was not able to find more detailed public documentation of this incident in Alberta, but mention is made in an article in a publication of the Oak Ridge National Laboratory, a federal research center associated with the Department of Energy, of a different unspecified incident on a drilling rig.
"Forces from methane hydrate dissociation have been blamed for a damaging shift in a drilling rig's foundation, causing a loss of $100 million," the article reports.Although public discussion of damage from methane hydrate accidents appears to be minimal, the danger is well-recognized within the industry. Last November, one Halliburton executive gave a presentation before a meeting of the American Association of Drilling Engineers in Houston, titled "Deepwater Cementing Consideration to Prevent Hydrate Destabilization."
It recognizes that the cementing process releases heat which can destabilize methane hydrates, and presents something called Cement System 2 as a solution to the problem. One of the graphs shows that the system doesn't achieve gel strength for four hours.
Yet according to an eyewitness report broadcast on Sunday on 60 Minutes, BP managers made the decision to decrease pressure in the well column by removing drilling mud before the cement had solidified in three plugs Halliburton had poured.
When a surge of gas started shooting up the well, a crucial seal on the blowout preventer at the well head on the ocean floor failed. It had been damaged weeks before and neglected as inconsequential by Transocean managers, according to the CBS news broadcast, even after chunks of rubber emerged from the drilling column on the surface.
According to the Associated Press, the victims of the Deepwater Horizon explosion said the blast occurred right after workers "introduced heat to set the cement seal around the wellhead." It is not known if Halliburton was employing Cement System 2, and testifying before the Senate last week, a Halliburton executive made no mention of methane hydrate hazards associated with cementing in deepwater.
A Promising Substance
Professors Koh and Sum are concerned that a focus on the dangers of methane hydrates in deepwater drilling will obscure their promise as an energy solution of the future. They are conducting research in the laboratory to create methane hydrates synthetically in order to take advantage of their peculiar properties. With their potential to store gas (both natural gas and hydrogen) efficiently within a crystalline structure, hydrogen hydrates could one day offer a potential solution for making fuel cells operate economically. Still at the fundamental stage, their work on storage is not yet complete enough to apply to commercial systems.
At the same time, there is an international competition underway to develop technology to harvest the vast deposits of methane hydrates in the world's oceans. Japan has joined the US and Canada in pursuit of this energy bonanza, motivated by the $23 billion it spends annually to import liquefied natural gas.
According to a Bloomberg News article called "Japan Mines Flammable Ice, Flirts with Environmental Disaster," the Japanese trade ministry is targeting 2016 to start commercial production, even as a Tokyo University scientist warned against causing a massive undersea landslide that could suddenly trigger a massive methane hydrate release.
The U.S. has a research program underway in collaboration with the oil industry, authorized by the Methane Hydrate Research and Development Act of 1999. The National Methane Hydrates R&D Program is housed at the National Energy Technology Laboratory (NETL) of the Department of Energy.
The National Academy of Sciences provided a briefing for Congress last January on the energy potential of methane hydrates based on its report which asserts that "no technical challenges have been identified as insurmountable" in the pursuit of commercial production of methane hydrates.
In the wake of the BP oil disaster, SolveClimate attempted to contact Dr. Charles Paull of the Monterey Bay Aquarium Research Institute, the lead author of the report. He was unavailable for comment, attending an international workshop on methane hydrates research in New Zealand from May 10-12, and according to his assistant, out of email contact.



This is agreat point to bring up.I offer the thoughts above as general inspiration but clearly there are questions like the oneyou bring up where the most important thing will beworking in honest good faith.